Vein disease treatments
VenaSeal™
procedure
Minimally invasive nonthermal treatment for vein disease and varicose veins.

The VenaSeal procedure delivers a small amount of a specially formulated medical adhesive to seal — or close — the diseased vein, rerouting blood to nearby healthy veins and providing symptom relief. The VenaSeal closure system is a safe and effective treatment, offering significant improvement in quality of life.1-4
The VenaSeal procedure
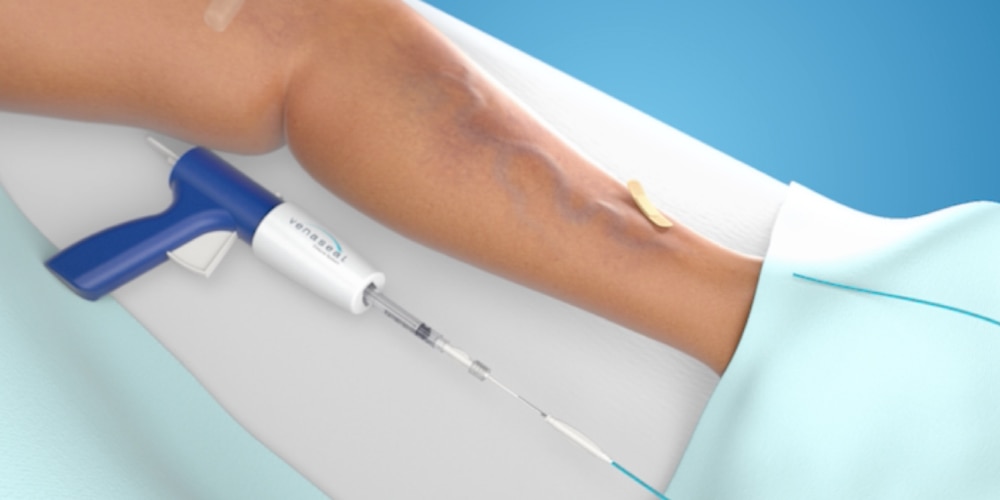
Step 1: Catheter is placed in the vein
Using an ultrasound, your doctor will position a catheter into the diseased vein through a small access site.
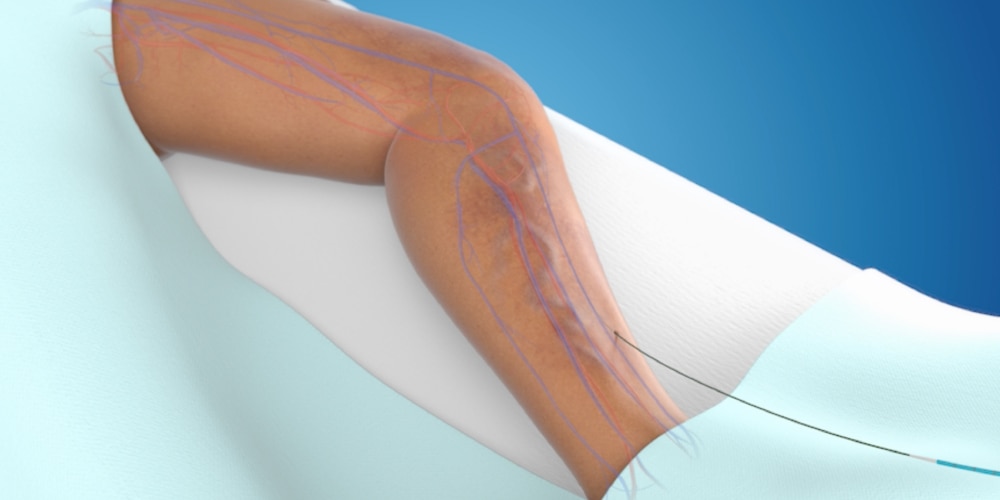
Step 2: Specialized adhesive is placed
A specially formulated medical adhesive is placed in the diseased vein via a small catheter.
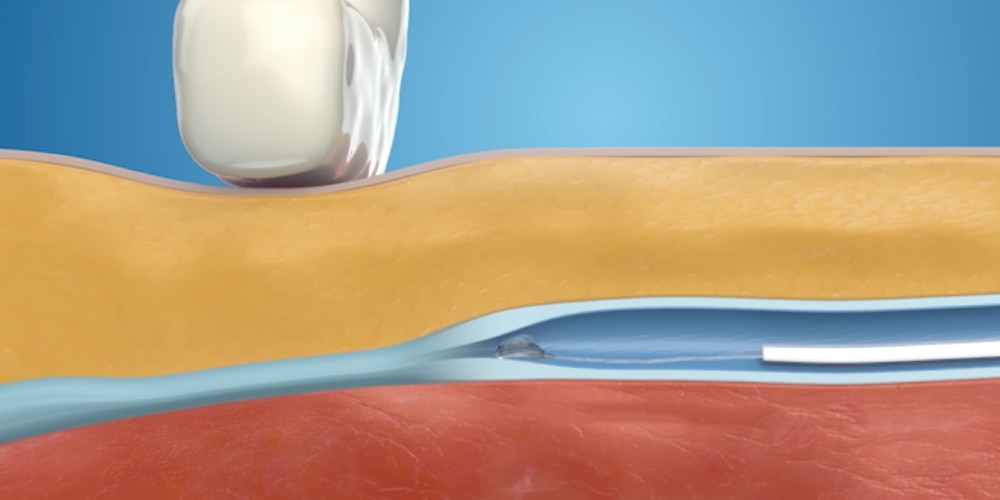
Step 3: Light external pressure is applied
Light external pressure is applied to adhere the vein walls together. This step is repeated for the length of the vein.
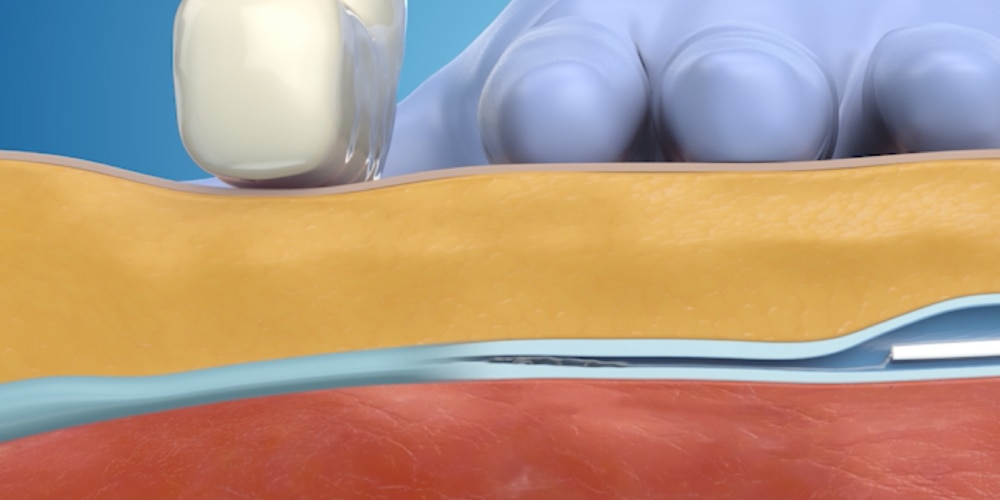
Step 4: Catheter is removed
The catheter is removed and a single adhesive bandage is applied to the vein access site.

I had all the signs and symptoms — tired legs, restless leg syndrome, achiness, the unsightly bulges. The treatment went very, very smoothly. I was very comfortable. I would suggest it to anybody.
– Heather A., VenaSeal patient
Before the procedure
You will have an ultrasound imaging exam of the leg being treated. This exam is important for assessing the diseased superficial vein and planning the procedure.
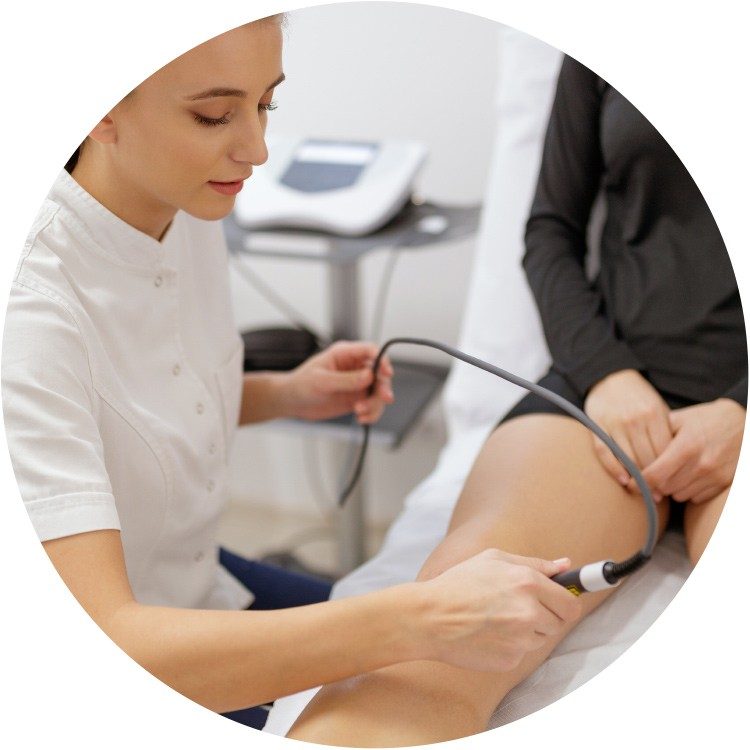
During the procedure
Your doctor can discuss the procedure with you. Here is a brief summary of what to expect:
- You may feel some minor pain or stinging with a needle stick to numb the site where the doctor will access your vein.
- Once the area is numb, your doctor will insert the catheter (i.e., a small hollow tube) into your leg. You may feel some pressure from the placement of the catheter.
- The catheter will be placed in specific areas along the diseased vein to deliver small amounts of the medical adhesive. You may feel a mild sensation of pulling. An ultrasound will be used during the procedure to guide and position the catheter.

After the procedure
- After treatment, the catheter is removed and a small adhesive bandage is placed over the puncture site.
- You will be taken to the recovery area to rest.
- Your doctor will recommend follow-up care as needed.

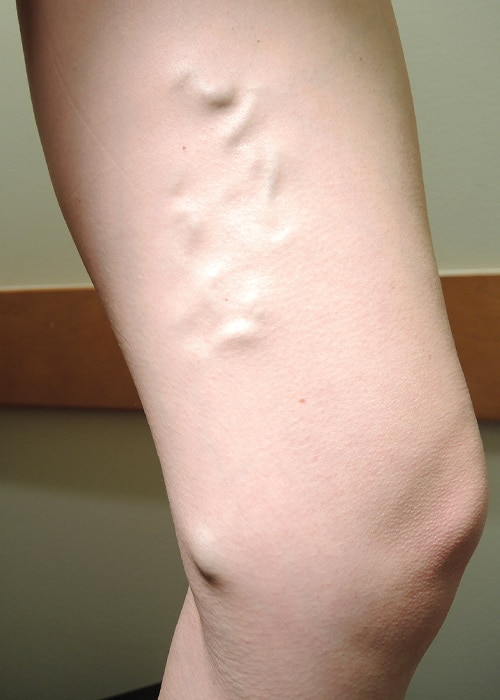
Patient results: before and after
Before VenaSeal procedure
Three months after VenaSeal procedure
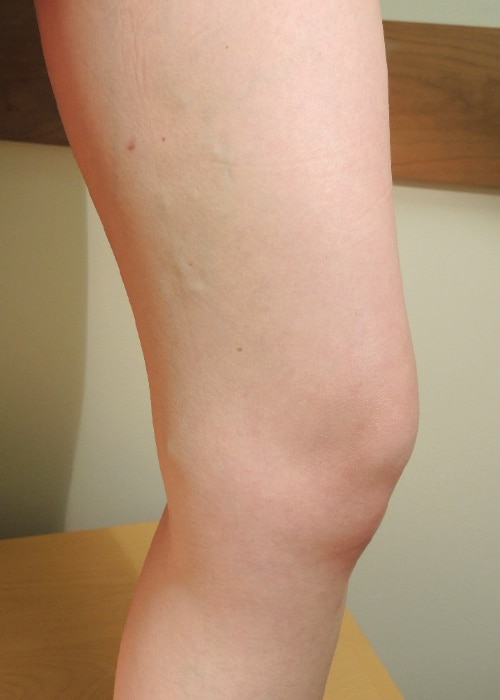
Individual results may vary. Images courtesy of Dr. Kathleen Gibson.
VenaSeal results and testimonials
The VenaSeal procedure went very smoothly; there was no pain involved.
– Heather A., VenaSeal patient
My leg heaviness is gone, and I can walk 18 holes of golf again.
– Steve H., VenaSeal patient
Benefits of the VenaSeal procedure
- Simple outpatient procedure
- Lasting results, with a 94.6% closure rate at five years1
- Compression stockings may not be needed after the procedure2,3
- Faster recovery time than thermal ablation1,2
- Less pain and bruising than thermal ablation1,2
- No tumescent anesthesia
Possible complications of the VenaSeal procedure may include allergic reaction, inflammation, phlebitis, deep vein thrombosis, and/or pulmonary embolism. Talk to your doctor about the risks and benefits.
Type of therapy
Closure with medical adhesive (permanent implant)
Inpatient or outpatient procedure
Outpatient procedure
One- versus two-leg treatment:
Typically, both legs can be treated in the same appointment
Needle sticks required
One needle stick for vein access
Typical post-treatment recovery
Typically limited to healing of vein access site; some patients may experience temporary skin and tissue irritation
Compression hose
Not required, but your doctor may recommend it post-procedure
Procedure success rate
94.6% closure rate at five years1
VenaSeal procedure FAQ
Is the VenaSeal procedure painful?
Most patients feel little, if any, pain during the outpatient procedure.1
Find a vein disease clinic near you.
References
Morrison N, Gibson K, McEnroe S, et al. Randomized trial comparing cyanoacrylate embolization and radiofrequency ablation for incompetent great saphenous veins (VeClose). J Vasc Surg. April 2015;61(4):985–994.
Proebstle T, Alm J, Dimitri S, et al. Three-year follow-up results of the prospective European Multicenter Cohort Study on Cyanoacrylate Embolization for treatment of refluxing great saphenous veins. J Vasc Surg Venous Lymphat Disord. March 2021;9(2):329–334.
Almeida JI, Javier JJ, Mackay EG, et al. Thirty-sixth-month follow-up of first-in-human use of cyanoacrylate adhesive for treatment of saphenous vein incompetence. J Vasc Surg Venous Lymphat Disord. September 2017;5(5):658–666.
Morrison N, Gibson, Vasquez M, et al. VeClose trial 12-month outcomes of cyanoacrylate closure versus radiofrequency ablation for incompetent great saphenous veins. J Vasc Surg Venous Lymphat Disord. May 2017;5(3):321–330.
Information on this site should not be used as a substitute for talking with your doctor. Always talk with your doctor about diagnosis and treatment information.