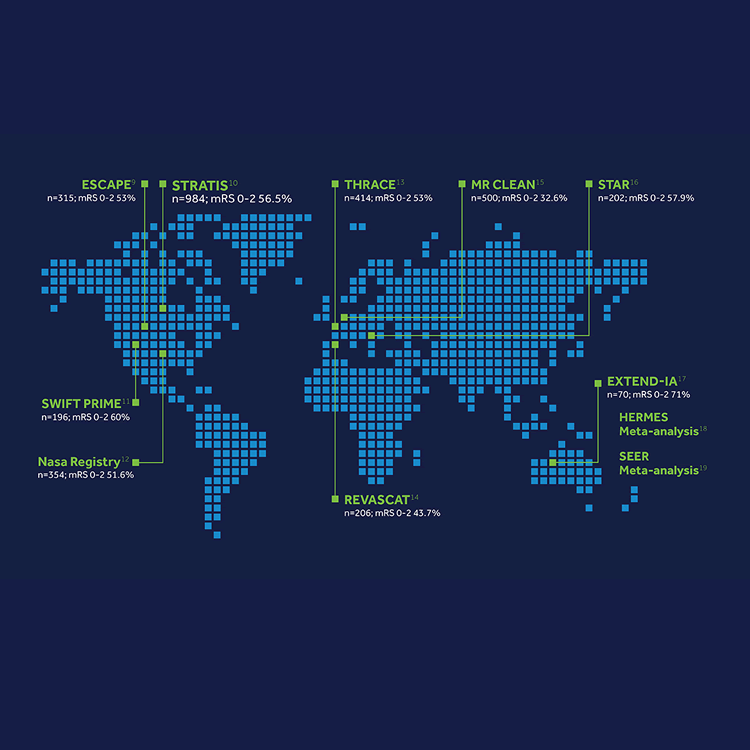

Robust data
for safety and efficacy
SWIFT PRIME
The SWIFT PRIME1 study was a global, multicenter, two-arm, prospective, randomized, open, blinded-endpoint (PROBE) trial to compare the functional outcomes in acute ischemic stroke (AIS) subjects treated with either IV t-PA alone or IV t-PA in combination with the Solitaire™ revascularization device.
Study conclusion
In patients receiving intravenous t-PA for acute ischemic stroke with confirmed large-vessel occlusions of the anterior circulation, treatment with the Solitaire™ stent thrombectomy device within 6 hours after symptom onset improved functional outcomes at 90 days.
EXTEND-IA
The EXTEND-IA2 trial was an investigator-initiated, multicenter, prospective, randomized, open, blinded-endpoint (PROBE) study in ischemic stroke patients receiving intravenous alteplase within 4.5 hours of stroke onset.
It was conducted to test the hypothesis that anterior circulation ischemic stroke patients, selected with a “dual target” of vessel occlusion and evidence of salvageable tissue on perfusion imaging within 4.5 hours of onset, would have improved reperfusion and early neurological improvement when treated with endovascular thrombectomy using the Solitaire™ FR stent thrombectomy device after intravenous (IV) alteplase, compared to alteplase alone.
Study conclusion
In ischemic stroke patients with a proximal cerebral arterial occlusion and salvageable tissue on CT perfusion imaging, early thrombectomy with the Solitaire™ FR stent thrombectomy device improves reperfusion, early neurologic recovery, and functional outcome compared with alteplase alone.
ESCAPE
The ESCAPE3 trial was an investigator-initiated, multi-center, prospective, randomized, open, blinded-endpoint (PROBE) trial designed to test whether patients with acute ischemic stroke, who were selected on the basis of results of CT and CTA, would benefit from rapid endovascular treatment involving contemporary endovascular techniques.
Patients were allocated 1:1 to endovascular treatment plus guideline-based care (intervention) vs. guideline-based care alone (control).
Study conclusion
Among acute ischemic stroke patients with proximal vessel occlusion, small infarct core, and moderate-to-good collaterals, rapid endovascular treatment improved functional outcomes and reduced mortality.
REVASCAT
REVASCAT4 was an investigator-initiated, multicenter, prospective, randomized, sequential, open,
It was conducted to determine the efficacy and safety of neurovascular thrombectomy with the Solitaire™ FR device in conjunction with medical therapy versus medical therapy alone, among eligible acute ischemic stroke patients treatable within 8 hours of symptom onset.
Study conclusion
In patients with anterior circulation stroke treatable within 8 hours of symptom onset, stent retriever thrombectomy reduces post-stroke disability and increases the rate of functional independence.
MR CLEAN
MR CLEAN5 was a randomized trial of intra-arterial treatment (IAT) for acute stroke. In patients with acute ischemic stroke caused by proximal intracranial arterial occlusion, IAT is highly effective for emergent revascularization. Five
Study conclusion
The MR CLEAN study observed that the addition of stent thrombectomy for acute ischemic stroke care is safe and effective when administered within 6 hours of symptom onset.
STRATIS
The STRATIS Registry6 was a prospective, multi-center, observational, single-arm study that was designed to investigate the ‘real-world’ stroke care questions and build a comprehensive representation of the patient population being treated with Medtronic stent retrievers.
The STRATIS Registry tracked the system of care data including transfer distances, referral patterns, times and location of stroke onset to ultimate interventional treatment. Patient images as well as techniques were independently core lab adjudicated. STRATIS is the first registry to enroll 984 patients within 8 hours from stroke onset with a focus on systems of care on clinical outcomes.
Study conclusion
Solitaire™ thrombectomy for large vessel ischemic stroke was safe and highly effective with substantially reduced disability. Benefits were consistent in all prespecified subgroups.
SWIFT PRIME- Saver JL, Goyal M, Bonafe A, et al. Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N. Engl. J. Med. Jun 11 2015; 372(24):2285-2295.
EXTEND-IA- Campbell BC, Mitchell PJ, Kleinig TJ, et al. Endovascular therapy for ischemic stroke with perfusion-imaging selection. N. Engl. J. Med. Mar 12 2015; 372(11):1009-1018.
ESCAPE- Goyal M, Demchuk AM, Menon BK, et al. Randomized assessment of rapid endovascular treatment of ischemic stroke. N. Engl. J. Med. Mar 12 2015; 372(11):1019-1030.
REVASCAT- Jovin TG, Chamorro A, Cobo E, et al. Thrombectomy within 8 hours after symptom onset in ischemic stroke. N. Engl. J. Med. Jun 11 2015; 372(24):2296-2306.
MR CLEAN- Berkhemer OA, Fransen PS, Beumer D, et al. A randomized trial of intraarterial treatment for acute ischemic stroke. N. Engl. J. Med. Jan 1 2015; 372(1):11-20.
STRATIS-Mueller-Kronast, NH, Zaidat O, et al. Systematic Evaluation of Patients Treated with Neurothrombectomy Devices for Acute Ischemic Stroke: Primary Results of the STRATIS Registry. Stroke 22 Aug 2017;48:2760–2768.
Disclaimer: This page may include information about products that may not be available in your region or country. Please consult the approved indications for use. Content on specific Medtronic products is not intended for users in markets that do not have authorization for use.
Important: Always refer to the Instructions For Use (IFU) packaged with the product/e-IFU for complete instructions, indications, contraindications, warnings, and precautions.
