Request more Information
Need support with developing your ICM pathway? Contact our team of market development specialists
Your browser is out of date
With an updated browser, you will have a better Medtronic website experience. Update my browser now.
Jeanette was 57 years old when she suffered a stroke. Hear how Reveal LINQ™ helped her find answers.
Since publication of the NICE DG41 Guidance, a group of clinicians has worked to create a guide to help overcome the challenges faced when implementing an ICM service. Read about their real-life experiences and learnings.
Download the guideThis report was developed with the support of the following people:
Reveal LINQ™ is recommended as an option to help detect atrial fibrillation after cryptogenic stroke, including transient ischaemic attacks (TIA), only if:
Read the full guidance: Visit the NICE website.
This webinar discusses the implications for NHS healthcare systems and healthcare providers based on the NICE recommendations. Key opinion leaders in the field of stroke care share their perspectives on the new Diagnostic Guidance and explain how these recommendations should guide use of ICMs in the treatment of stroke patients.
Key learning objectives:
Presented by: Dr Klaus Witte, Dr Matthew Reynolds, Dr Paul Guyler
In line with the NICE Diagnostics Guidance 41, Salford Royal NHS Foundation Trust added the use of an implantable cardiac monitor in cryptogenic stroke patients to their post-stroke aftercare pathways to help increase the detection of Atrial fibrillation (AF).
Introducing implantable cardiac monitors to support detection of atrial fibrillation after cryptogenic stroke at Salford Royal NHS Trust.
Reveal LINQTM with TruRhythmTM enables higher AF detection and lower stroke recurrence associated with Stroke patients versus standard of care.1

Additional ECG monitoring by long-term non-invasive ECG monitors or ICMs should be considered to document silent AF and prevent secondary stroke.10
Lowest published rate of AF false episodes12-15
Disclaimer: A controlled, head-to-head study evaluating the comparative performance of these devices has not been done.
*Based on AF episodes ≥ 2 minutes and in known AF patients. % of false positives = (1 – episode PPV). AF episodes PPV may vary between gross and patient average.
†Confirm™ Rx with SharpSense™ technology & BIOMONITOR III have no published clinical evidence showing AF episode PPV or AF sensitivity.
In a known AF patient population. % Full-time Employee 100 Patient Clinic, 40 hours per week, 44 weeks/year.
In addition to the recent conclusion of the NICE Diagnostic Assessment Programme that Reveal LINQ™ is likely a cost-effective use of NHS resources28, multiple other studies have also demonstrated ICM cost effectiveness .
*A measure of the state of health of a person or group in which the benefits, in terms of length of life, are adjusted to reflect the quality of life. One QALY is equal to 1 year of life in perfect health.
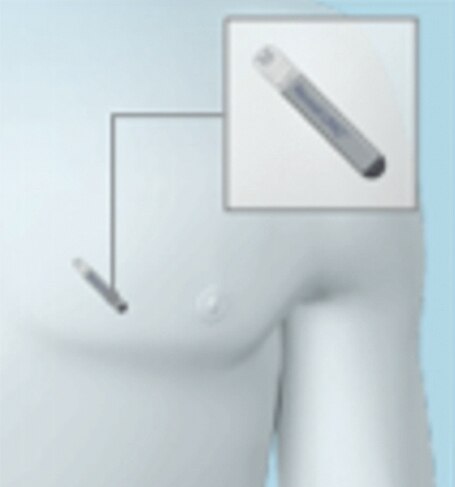
The Reveal LINQ™ Insertable Cardiac Monitor (ICM) is placed just under the skin of the patient’s chest. Key benefits include:
100% of patients found Reveal LINQTM ICM did not limit their activities of daily living
Insertion procedure is minimally invasive and brief
Not visible in most patients.
Safe for MRI at 1.5 and 3.0T even on the same day of insertion.9
The implant of a loop recorder is not surgery - I call it an injection that is going to enable us to watch every heartbeat, every day for three years, to look for one of the most common causes of a cryptogenic stroke.
Dr Don Sawyer, Electrophysiologist
Tsivgoulis G, et al. Prolonged Cardiac Rhythm Monitoring and Secondary Stroke Prevention in Patients With Cryptogenic Cerebral Ischemia. Stroke. Published online June 20, 2019. same as reference
Sanna T, et al. N Engl J Med. 2014;370:2478-2486.
Wolf PA, et al. Arch Intern Med. 1987;147:1561-1564.
Lin HJ, et al. Stroke. 1996; 27:1760-1764
ICM Size Comparison Guide. Medtronic data on file. 2019.
ICM Accuracy Comparison Guide. Medtronic data on file. 2019.
TruRhythm Detection Algorithms. Medtronic data on file. 2017.
Rogers, John D., et al. Journal of Medical Economics (2020): 1-8. 22. Pürerfellner H, et al. Europace. 2018;20:f321-f328
Reference the Reveal LINQ ICM Clinician Manual for usage parameters.
Kirchhof P, Benussi S, Kotecha D, et al. “2016 ESC Guidelines for the management of atrial brillation developed in collaboration with EACTS.” EurHeart J.October 7, 2016;37(38):2893-2962.
Kolominsky-Rabas PL, Heuschmann PU, Marschall D, et al. Lifetime cost of ischemic stroke in Germany: results and national projections from a population based registry
Pürerfellner H, et al. Europace. 2018;20:f321-f328.
Confirm Rx™ ICM DM3500 FDA Clearance Letter. 2017.
Nölker G, et al. J Cardiovasc Electrophysiol. 2016;27:1403-1410.
Biotronik BioMonitor 2 Technical Manual. 2017.
Nölker G, Mayer J, Boldt LH, et al. Performance of an Implantable Cardiac Monitor to Detect Atrial Fibrillation: Results of the DETECT AF Study. J Cardiovasc Electrophysiol. December 2016;27(12):1403-1410.
Diamantopoulos A, et al. Journal of Stroke. 2016: 0(0): 1-11.
Edwards J, et al. Journal of the American Heart Association. 2020;9:e013227. DOI:10.1161/JAHA. 119.013227
Witte K.K., et al. Value in Health (ISPOR). 2018: 3(21): 258
Thijs V, et al. Journal of Neurology, Neurosurgery & Psychiatry. 2018;89;A6
Chew d, et al. Journal of the American College of Cardiology. 2020;75(11) 315
Sawyer L, et al. Cost-effectiveness of Insertable Cardiac Monitors to Identify Atrial Fibrillation After Cryptogenic Stroke in the United States. Congress Poster – HRS 2020, May
Reynolds M, et al. Value in Health (ISPOR) 2020:1(23).94-95
Rogers, John D., et al. Journal of Medical Economics (2020): 1-8
Kipp R, et al.PACE. 2017;40:982-985.
Kanters, Tim A., et al. Europace (2015): euv217.
Wong GR, et al. Int J Cardiol. 2016;223:13-17.
NICE Diagnostics Guidance 41: Implantable cardiac monitors to detect atrial fibrillation after cryptogenic stroke. September 2020 © NICE 2020. View full guidance.
Buck B. et al. “PERDIEM Post-Embolic Rhythm Detection with Implantable versus External Monitoring Trial: Final Results.” Oral presentation at the 5th European Stroke Organization Conference (ESOC), Milan, Italy, May 2019.