Tissue valves and conduits
Mosaic™ mitral bioprosthesis
Tissue valves and conduits
Mosaic™ mitral bioprosthesis
The Mosaic™ mitral bioprosthesis is a next-generation native porcine valve for mitral valve replacement.
Features
Built for a life. Time tested.
Based on more than 50 years of Medtronic tissue-valve design expertise, the Mosaic™ mitral valve is a great option for surgeons seeking unsurpassed long-term durability, excellent hemodynamics, and a smooth implanting experience.1–3
Durability
Published clinical experience demonstrates industry-leading long-term performance.1,3
Features that may contribute to the Mosaic™ mitral valve’s durability are:
- Proprietary amino oleic acid (AOA™) anti-calcification tissue treatment with clinical use across a suite of devices in more than half a million patients for over 30 years†
- High-performance stent that allows for absorption of stress produced during the cardiac cycle4
- Physiologic fixation, our advanced tissue fixation process, to mitigate biomechanical failures and promote long-term valve durability by:
- Improving preservation of valve structure and leaflet function, allowing it to function similar to a native valve5,6
- Allowing leaflets to remain soft and flexible, which protects the tissue from cyclic fatigue5,6
Learn more about the Mosaic™ mitral valve’s unsurpassed durability. (03:27)
AOA™ anti-calcification treatment
The Medtronic-patented AOA™ tissue treatment is designed to reduce calcification and protect the tissue.†
- AOA™ covalently bonds with free aldehydes and washes lipids away.7–9
- Large AOA™ molecules slow diffusion of calcium into tissue matrix.
Learn more about mitigating calcification with AOA™ tissue treatment. (03:31)
Physiologic tissue fixation
- Leaflets float at net-zero pressure.
- Roots are pressurized at 40 mmHg with glutaraldehyde.
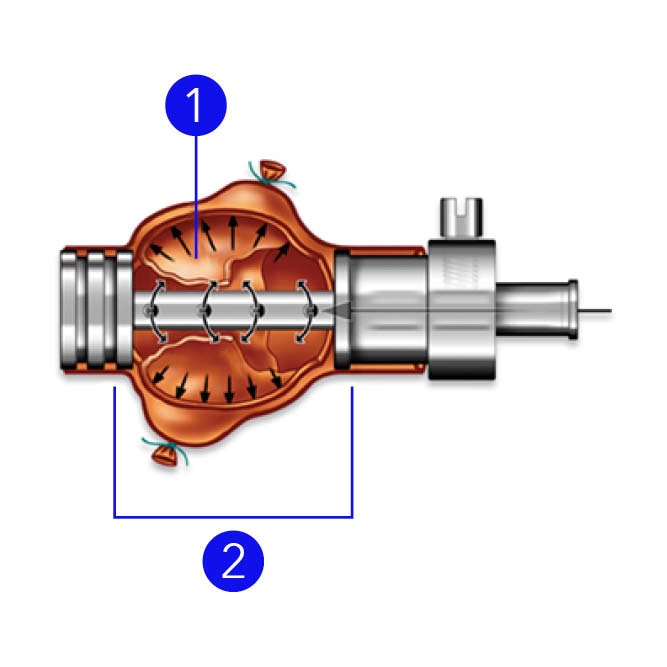
Hemodynamics
The Mosaic™ mitral valve was designed to allow for maximum blood flow. The creation of the valve — from the porcine leaflets and valve geometry (specifically stent height) to how the tissue is mounted in the frame — affects how optimally the blood flows in diastole, and how well the leaflets coapt in systole.
As reported in the Mosaic™ mitral valve instructions for use, hemodynamic results at one year2:
| Mosaic™ mitral valve size3 | 25 | 27 | 29 | 31 | 33 |
|---|---|---|---|---|---|
| Mean gradient (mm) (n = ) |
5.7 ± 1.7 (41) | 4.6 ± 2.1 (98) | 4.4 ± 1.8 (123) | 3.7 ± 1.4 (50) | 3.4 ± 1.8 (8) |
| Mean effective orifice area (cm2) (n = ) |
1.6 ± 0.4 (35) | 1.7 ± 0.5 (90) | 1.8 ± 0.5 (114) | 1.7 ± 0.5 (43) | 1.9 ± 0.5 (6) |
Geometry and reintervention
Designed to accommodate ventricular flow
The Mosaic™ mitral valve reflects the asymmetry of the native porcine valve, providing one wider leaflet spaced at 135 degrees. This largest leaflet of the Mosaic™ mitral valve is intended to align with the patient’s anterior mitral leaflet to accommodate ventricular flow.
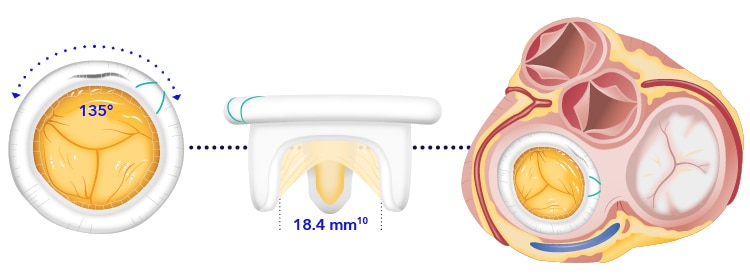
Suitable for future interventions
Valve dimensions and geometry facilitate future valve-in-valve (ViV) procedures.
- Radiopaque stent post eyelets on the Mosaic™ mitral valve provide visible markers to help correctly position the transcatheter valve during ViV procedures.
- MR Conditional, nonmetallic frame mitigates risk of corrosion between surgical valve and transcatheter valve stent materials.
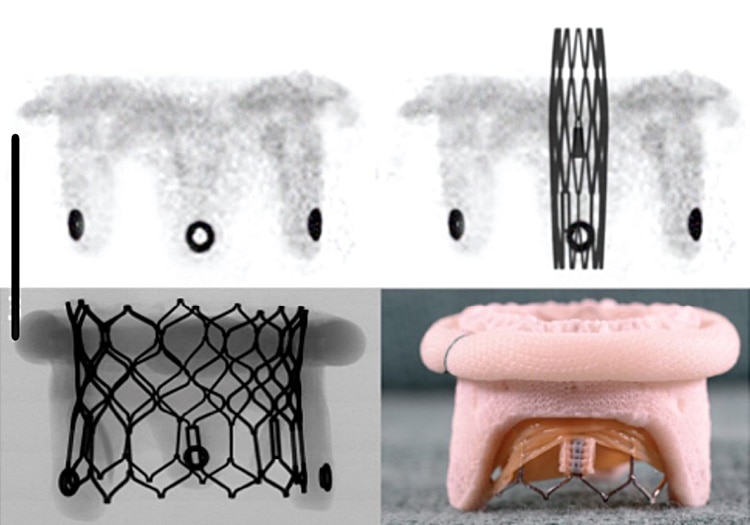
SAPIEN™* device deployed inside Mosaic™ mitral valve
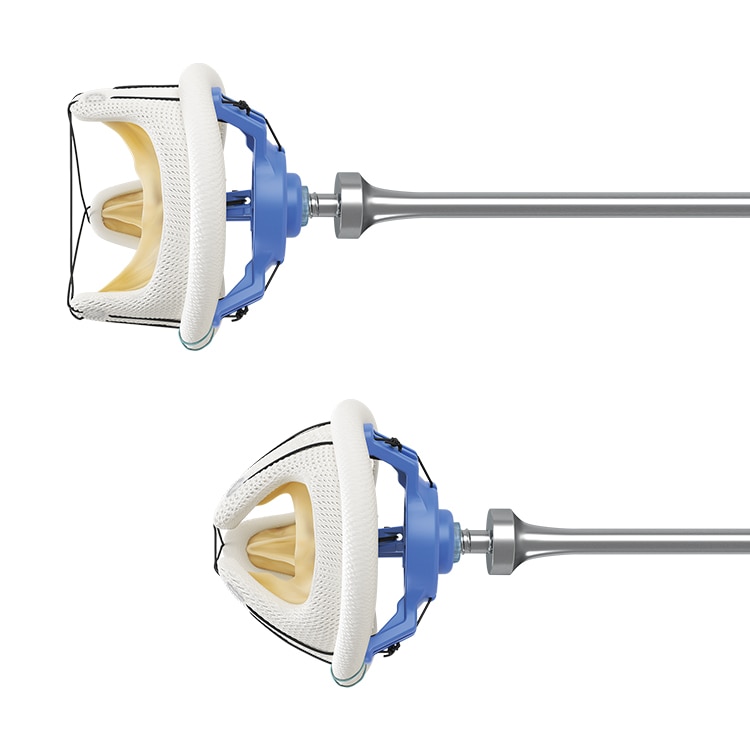
Implantability
The Cinch™ implant system capitalizes on the flexible stent and offers different features to facilitate valve implantation:
- Aids minimally invasive procedures through small incisions11
- Helps prevent suture looping11
- Designed to protect tissue from inadvertent damage and prevent entanglements with the sub-valvular apparatus
- Assists implantation with clear marking for proper orientation2
- Offers smooth needle penetration and suture placement with a pliable, conformable cuff12
Clinical evidence
Long-term outcomes of mitral valve replacements
Mosaic™ mitral valve versus Carpentier-Edwards bovine pericardial mitral valves
This retrospective study compared the long-term outcomes of the Mosaic™ porcine mitral valve to Carpentier-Edwards bovine pericardial mitral valves in 940 patients.1
Cumulative incidence of reoperation at 15 years (p < 0.001):
- 7.9% for Mosaic™ mitral valve
- 13.2% for CE Perimount™* or Magna™* mitral valve
Survival at 15 years (p = 0.67):
- 24.0% for Mosaic™ mitral valve
- 16.5% for CE Perimount™* or Magna™* mitral valve
Mosaic™ mitral valve
CE Perimount™*/Magna™* mitral valve
On average, the time before reoperation for structural valve deterioration (SVD) was 4.3 years longer with the Mosaic™ porcine valve than Carpentier-Edwards pericardial valves.
Time to reoperation for SVD‡
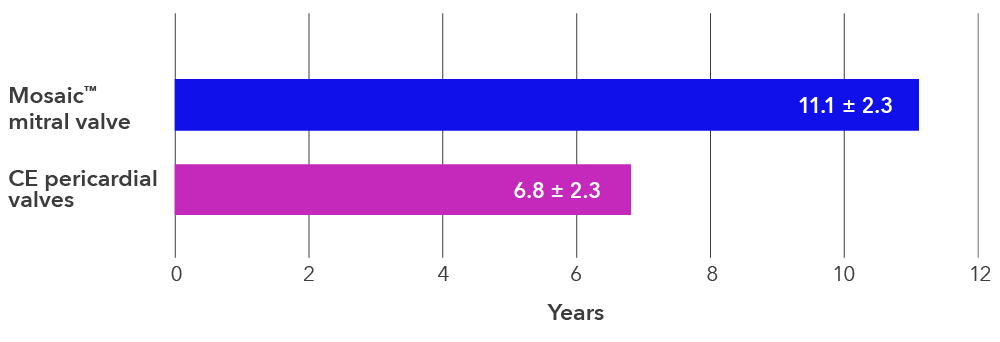
Mosaic™ mitral valve versus Epic™* mitral valve
This retrospective, single-center study analyzed long-term outcomes of 335 patients undergoing isolated mitral valve replacement.13
Freedom from reintervention at 10 years (p = 0.1):
- 79.1% for Mosaic™ mitral valve
- 62.2% for Epic™* mitral valve
Freedom from definite (confirmed on reoperation) SVD (p = 1.00):
- 91.7% for Mosaic™ mitral valve group
- 89.4% for Epic™* mitral valve group
Mosaic™ mitral valve
Epic™* mitral valve
Cumulative incidence of reintervention with death as competing risk
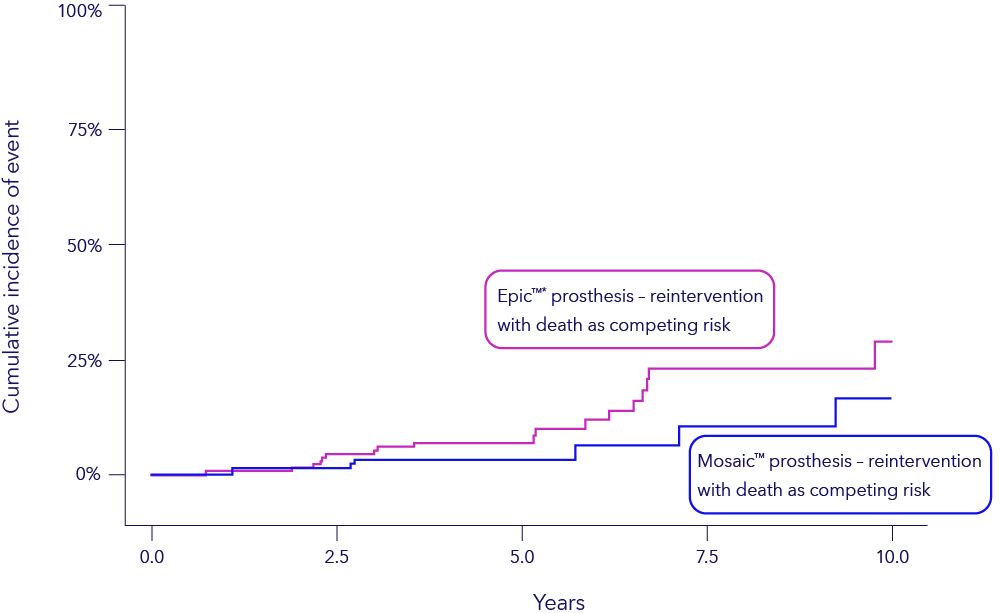
“At 10 years after mitral valve replacement, the cumulative incidence of reintervention was twice as high in the Epic™* group.”§
Ordering information
Mosaic™ mitral valve, Model 310
 |
A: Valve size |
| B: Orifice diameter |
|
| C: Suture ring diameter | |
| D: Valve height | |
| E: Ventricular protrusion |
| Item number | A: Valve size (stent OD◊) (± 0.5 mm) |
B: Orifice diameter (stent ID) (± 0.5 mm) |
C: Suture ring diameter (± 1 mm) |
D: Valve height (± 0.5 mm) |
E: Ventricular protrusion (± 0.5 mm) |
|---|---|---|---|---|---|
| 310C25 | 25 | 22.5 | 33.0 | 18.0 | 13.5 |
| 310C27 | 27 | 24.0 | 35.0 | 19.0 | 14.0 |
| 310C29 | 29 | 26.0 | 38.0 | 20.5 | 15.5 |
| 310C31 | 31 | 28.0 | 41.0 | 22.0 | 17.0 |
| 310C33 | 33 | 30.0 | 43.0 | 23.0 | 17.5 |
Accessories
| Item number | Description |
|---|---|
| T7615MSM | Tray, accessory, Mosaic™ mitral |
| 7639 | Handle (234 mm length) pliant, without locknut to be used with Mosaic™ or Mosaic Ultra™ prostheses |
| 7639XL | Handle (368 mm length) pliant, without locknut to be used with Mosaic™ or Mosaic Ultra™ prostheses |
| 7310 | Mosaic™ mitral obturator set (no handles, no tray) |
Resources
Similar products
-
 Avalus Ultra™ bioprosthesis
Avalus Ultra™ bioprosthesisAvalus Ultra™ bioprosthesis: A next-generation bovine pericardial valve for aortic valve replacement.
-
 CG Future™ annuloplasty ring and band
CG Future™ annuloplasty ring and bandThe CG Future™ annuloplasty ring and band provide targeted remodeling for mitral valve repair.
-
 SimuForm™ annuloplasty ring
SimuForm™ annuloplasty ringThe SimuForm™ annuloplasty ring has a flexible segment designed to accommodate anterior motion, while the semi-rigid posterior segment enables remodeling.
-
 SimuPlus™ flexible annuloplasty ring and band
SimuPlus™ flexible annuloplasty ring and bandThe SimuPlus™ flexible annuloplasty ring and band offer flexible solutions to mitral and tricuspid (band only) valve repair.
TM* Third-party brands are trademarks of their respective owners.
† The benefits of AOA™ tissue treatment have been demonstrated through animal testing. No direct clinical evaluation of the benefits of AOA™ treatment in humans has been conducted.
‡ SVD was defined, according to STS, as dysfunction or deterioration involving the operated valve, exclusive of infection or thrombosis, as determined by reoperation, autopsy, or clinical investigation. Mean follow-up times were significantly different (Mosaic™: 7.0 ± 4.8 versus Edwards: 6.0 ± 3.9, p = 0.002).
§ 95% CI 32.7%–36.1% in the Epic™* group; 16.2%–18.9% in the Mosaic™ group.
◊ Equivalent to annulus diameter.
- Beute T, Goehler M, Parker J, et al. Long-term outcomes of Mosaic versus Perimount mitral replacements: 17-year follow up of 940 implants. Ann Thorac Surg. 2020;110(2):508–515. doi: 10.1016/j.athoracsur.2019.10.075.
- Mosaic™ porcine bioprosthesis instructions for use. Medtronic, Inc. 1220016001 rev. 1C; 2018.
- Rieß FC, Fradet G, Lavoie A, Legget M. Long-term outcomes of Mosaic bioprosthesis. Ann Thorac Surg. 2018;105(3):763–769. doi: 10.1016/j.athoracsur.2017.09.053.
- Reis R, Hancock WD, Yarbrough JW, Glancy DL, Morrow AG. The flexible stent. A new concept in the fabrication of tissue heart valve prostheses. J Thorac Cardiovasc Surg. 1971;62(5):683–689. passim.
- Vesely I. Analysis of the Medtronic Intact bioprosthetic valve. Effects of “zero-pressure“ fixation. J Thorac Cardiovasc Surg. 1991;101(1):90–99.
- Mayne ASD, Christie GW, Smaill BH, Hunter PJ, Barratt-Boyes BG. An assessment of the mechanical properties of leaflets from four second-generation porcine bioprostheses with biaxial testing techniques. J Thorac Cardiovasc Surg. 1989;98(2):170–180.
- Girardot MN, Girardot JM, Schoen FJ. Development of the AOA process as antimineralization treatment for bioprosthetic heart valves. Trans Soc Biomat. 1993;53(2):207–215; discussion 216.
- Girardot MN, Torrianni M, Girardot JM. Effect of AOA on glutaraldehyde-fixed bioprosthetic heart valve cusps and walls: binding and calcification studies. Int J Artif Organs. 1994;17(2):76–82.
- Gott JP, Pan-Chih, Dorsey JM, et al. Calcification of porcine valves: a successful new method of antimineralization. Ann Thorac Surg. 1992;53(2):207–215. doi: 10.1016/0003-4975(92)91321-y.
- Based on internal testing report D00807486_A. Information shown on this page represents measurement for size 29 mm Mosaic™ mitral valve. July 28, 2022.
- Takagi K, Arinaga K, Takaseya T, et al. Hemodynamic and clinical performance of the 25-mm Medtronic Mosaic porcine bioprosthesis in the mitral position. J Artif Organs. 2021;25(1):34–41. doi: 10.1007/s10047-021-01277-1.
- Based on human factors engineering report, 10137288DOC rev 1A surg; 98:170–180. 1998.
- Tomšič A, de Weger A, van der Stoel M, Klautz RJM, Palmen M. A nationwide study on mitral valve repair vs replacement for active endocarditis. Ann Thorac Surg. 2024;117(1):120–126. doi: 10.1016/j.athoracsur.2023.08.032.





