Indications, Safety, and Warnings
Ablation products for atrial fibrillation
To view the indications, safety, and warnings, scroll down or click on one of the links below to jump you down the page to that product.
Achieve Advance mapping catheter
Indications
The Achieve Advance mapping catheter is indicated for multiple electrode electrophysiological mapping of the cardiac structures of the heart, i.e., recording or stimulation only. The Achieve mapping catheter is designed to obtain electrograms in the atrial regions of the heart.
Contraindications
The catheter is contraindicated as follows:
- For use as an ablation device
- For use with transseptal sheaths featuring side holes larger than 1.00 mm (0.04 in) in diameter
- Retrograde approach
Electrophysiology studies are contraindicated when the patient’s underlying cardiac disease makes it likely that induced arrhythmias will be extremely difficult to terminate and carry a high risk of death, as in the following conditions:
- An active systemic infection
- Left atrial thrombus
- Pulmonary vein stents
- Prosthetic heart valve (tissue or mechanical)
- Myxoma
- Interatrial baffle or patch
- Conditions where the manipulation of the catheter within the heart would be unsafe
- Acute myocardial infarction
See the device manual for detailed information regarding the procedure, indications, contraindications, warnings, precautions, and potential complications/adverse events.
Caution: Federal law (USA) restricts this device to sale by or on the order of a physician.
Back to top
Achieve mapping catheter
Indications
The Achieve mapping catheter is indicated for multiple electrode electrophysiological mapping of the cardiac structures of the heart, i.e., recording or stimulation only. The Achieve Mapping Catheter is designed to obtain electrograms in the atrial regions of the heart.
Contraindications
The catheter is contraindicated as follows:
- For use as an ablation device
- For use with transseptal sheaths featuring side holes larger than 1.00 mm in diameter
- Retrograde approach
Electrophysiology studies are contraindicated when the patient’s underlying cardiac disease makes it likely that induced arrhythmias will be extremely difficult to terminate and carry a high risk of death, as in the following conditions:
- An active systemic infection
- Left atrial thrombus
- Pulmonary vein stents
- Prosthetic heart valve (tissue or mechanical)
- Myxoma
- Interatrial baffle or patch
- Conditions where the manipulation of the catheter within the heart would be unsafe
- Acute myocardial infarction
Refer to the device manual for detailed information regarding the procedure, indications, contraindications, warnings, precautions, and potential complications/adverse events. For further information, please call Medtronic at 800-238-2518 and/or consult the Medtronic website at medtronic.com.
Back to top
Arctic Front family of cardiac cryoablation catheters
Indications
The Arctic Front family of cardiac cryoablation catheter systems is indicated for the treatment of drug refractory recurrent symptomatic paroxysmal and persistent atrial fibrillation (episode duration less than six months). The Arctic Front Advance and Arctic Front Advance Pro Cardiac Cryoablation Catheters are also indicated for the treatment of recurrent, symptomatic, and paroxysmal atrial fibrillation as an alternative to anti-arrhythmic drug therapy as an initial rhythm control strategy.
Contraindications
Use of the cryoballoon is contraindicated: 1) In the ventricle because of the danger of catheter entrapment in the chordae tendineae, 2) In patients with one or more pulmonary vein stents, 3) In patients with cryoglobulinemia, 4) In patients with active systemic infections, and 5) In conditions where the manipulation of the catheter within the heart would be unsafe (e.g., intracardiac mural thrombus).
Warnings/Precautions
Do not reprocess or resterilize this device for the purpose of reuse. Administer appropriate levels of peri-procedural anticoagulation therapy for patients undergoing left-sided and transseptal cardiac procedures. The Arctic Front family of Cryoballoons have not been studied for the safety of changes in anticoagulation therapy in patients with paroxysmal atrial fibrillation. Do not inflate the balloon inside the sheath or while the catheter is positioned inside a pulmonary vein. Always inflate the balloon in the atrium and then position it at the pulmonary vein ostium. Atrial Fibrillation patients with significant left ventricular dysfunction, advanced heart failure, severe left atrial enlargement, or significant structural heart disease were excluded from the clinical trials that supported the approved indications. For baseline demographics and clinical characteristics of the patients studied, please see the Clinical Summaries listed in the IFU. If the balloon cannot be inflated or deflated using the CryoConsole, have a Manual Retraction Kit on hand during the procedure. (Refer to the CryoConsole Operator’s Manual for more detailed instructions on the Manual Retraction Kit). Disconnect the catheter’s electrical connection before cardioversion or defibrillation.
Catheter handling:
- Use extreme care when manipulating the catheter.
- Do not use excessive force to advance, withdraw, or rotate the catheter, especially if resistance is encountered.
- Do not use the catheter if it is kinked, damaged, or cannot be straightened.
- If the catheter becomes kinked or damaged while in the patient, remove it and use a new catheter.
- Straighten the shaft before inserting or withdrawing the catheter.
- Do not at any time preshape or bend the catheter shaft or balloon segment.
- Catheter advancement should be performed using fluoroscopy or other appropriate techniques.
- Do not position the cryoballoon catheter within the tubular portion of the pulmonary vein.
Do not advance the balloon beyond the guide wire or circular mapping catheter to reduce the risk of tissue damage. Ensure that the guide wire or circular mapping catheter is inserted into the catheter and through the balloon portion for adequate support during vascular access insertion. Do not pass the catheter through a prosthetic heart valve (mechanical or tissue). Do not pull on the balloon catheter, circular mapping catheter, sheath, umbilical cables, or CryoConsole while the balloon catheter or circular mapping catheter are frozen to tissue. Before moving these components, use appropriate techniques to ensure that the balloon catheter and circular mapping catheter are not adhered to tissue.
Always advance and withdraw components slowly to minimize the vacuum created and therefore minimize the risk of air embolism. Do not expose the catheter handle or coaxial and electrical connectors to fluids or solvents. The use of fluoroscopy during catheter ablation procedures presents the potential for significant X-ray exposure to both patients and laboratory staff. Give careful consideration before using the device in pregnant women. Flush the guide wire lumen before initial insertion and then frequently throughout the procedure to prevent coagulation of blood in the lumen. Flush the guide wire lumen with saline after each contrast injection. Use only 0.081 cm (0.032 in) or 0.089 cm (0.035 in) guide wires with the catheter. Do not connect the cryoablation catheter to a radiofrequency (RF) generator or use it to deliver RF energy. Use only isolated equipment (IEC 60601-1 Type CF equipment, or equivalent) with the CryoConsole and catheters. Avoid catheter entanglement with other catheters, devices, or wires.
Before powering up an RF generator or applying RF energy, disconnect the cryoablation catheter from the CryoConsole. Always deflate the balloon and withdraw the balloon into the transseptal sheath before removing the balloon from the left atrium. If the sterile packaging or catheter is damaged, do not use the catheter. Use only Medtronic cryoablation catheters, 12 FR inner diameter sheaths, circular mapping catheters, refrigerant tanks, and components with the CryoConsole. Closely monitor patients undergoing cardiac ablation procedures during the post-ablation period for clinical adverse events. This cryoablation system should be performed only in a fully equipped facility under the supervision of physicians trained in cryoablation procedures.
Potential Complications
Potential complications/adverse events from cardiac catheterization and ablation include, but are not limited to, the following: Access site complications (e.g., bruising, ecchymosis); Anemia; Anxiety; Arrhythmia (e.g., atrial flutter, bradycardia, heart block, tachycardia); Back pain; Bleeding from puncture sites; Bronchial constriction; Bronchial fistula; Bronchitis; Bruising; Cardiac tamponade; Cardiopulmonary arrest; Cerebral vascular accident; Chest discomfort/pain/pressure; Cold feeling; Coronary artery spasm; Cough; Death; Diarrhea; Dizziness; Embolism; Esophageal damage (including esophageal fistula); Fatigue; Fever; Headache; Hemoptysis; Hypotension/Hypertension; Infection (e.g., pericarditis, sepsis, urinary); Lightheadedness; Myocardial infarction; Nausea/vomiting; Perforation; Pericardial effusion; Phrenic nerve injury; Pleural effusion; Pneumonia; Pneumothorax; Pseudoaneurysm; Pulmonary edema; Pulmonary hemorrhage; Pulmonary vein dissection; Pulmonary vein stenosis; Shivering; Shortness of breath; Sore throat; Transient ischemic attack; Vagal nerve injury (e.g., gastroparesis); Vasovagal reaction; Visual Changes (e.g., blurred vision).
Refer to the device technical manual for detailed information regarding the procedure, indications, contraindications, warnings, precautions, and potential complications/adverse events. For further information, please call Medtronic at 800-238-2518 and/or consult the Medtronic website at medtronic.com.
Caution: Federal law (USA) restricts this device to sale by or on the order of a physician.
Back to top
DiamondTemp ablation system
Indications (or Intended Use)
The DiamondTemp catheter is indicated for use in cardiac electrophysiological mapping (stimulation and recording) and for treatment of drug refractory, recurrent, symptomatic paroxysmal atrial fibrillation when used in conjunction with the DiamondTemp RF generator and accessories (DiamondTemp catheter-to-RF generator cable, DiamondTemp GenConnect cable, DiamondTemp EGM cable, DiamondTemp irrigation pump, DiamondTemp irrigation tubing set) and compatible mapping system.
Contraindications
Use of the DiamondTemp catheter is contraindicated for: 1) patients with active systemic infection, 2) patients with prosthetic valves, 3) patients with intracardiac thrombus or myxoma, or interatrial baffle or patch via transseptal approach, 4) patients unable to receive heparin or an acceptable alternative to achieve adequate anticoagulation, 5) pregnant women and children <18 years of age, and 6) patients who are hemodynamically unstable.
Warnings and Precautions
Cardiac ablation procedures should be performed only by physicians trained in the techniques of RF catheter ablation in a fully equipped electrophysiology (EP) laboratory. The catheter is for single use only. Do not reprocess or resterilize. Reusing, reprocessing, or resterilizing may compromise the structural integrity of the device or lead to product failure, which may result in patient injury, illness, or death. Reuse, reprocessing, or resterilizing may also create a risk of contamination of the device. Contamination may lead to injury, illness, or death of the patient. Pacemakers, implantable cardioverter defibrillators (ICDs), and leads can be adversely affected by RF signals. ICDs should be deactivated prior to ablation, precautions should be taken when the catheter is in close proximity to leads, and complete system analysis should be performed after ablation. Long-term risks of RF ablation lesions have not been established. Ablation too close to the esophageal area can result in esophageal fistula. Ablation near the AV node can cause permanent or partial conduction block. To ensure proper operation of the tissue contact impedance measurement function, all four electrodes and six thermocouples on the catheter tip must protrude from the distal tip of the guiding sheath. Carefully monitor the tissue contact impedance before delivery of RF energy. Do not place the RF electrode in proximity to any other mapping or ablation electrodes, as this may cause inadvertent, ineffective, or unsafe tissue ablation and may increase chances of char, coagulum, or steam pops. Although a higher contact impedance value typically indicates acceptable tissue contact, and low contact impedance values typically indicate lack of tissue contact, caution should be exercised. Areas of previously ablated tissue may also display a low contact impedance value. Other parameters, such as EGM, fluoroscopic images, and intracardiac ultrasound should be monitored before deciding to apply RF. Stimulation of cardiac tissues caused by pacing stimulus or RF energy may lead to inadvertent induction of arrhythmias. These arrhythmias may require defibrillation that could also result in skin burns. Do not use the catheter for epicardial ablation. Using ablation parameters (such as temperature set-point, ablation duration, or irrigation flow rate) other than those recommended by Medtronic may be hazardous to patients. Exercise caution and sound medical reasoning when deciding to deviate from recommended parameters. Perform catheter advancement under fluoroscopic guidance in conjunction with internal contact, electrograms, and impedance monitoring to minimize the risk of cardiac damage, perforation, or tamponade. Tip-to-tissue contact impedance is actively monitored only before and after ablation. During ablation, use caution when the temperature drops suddenly. A drop in temperature may be associated with loss of tissue contact. In case of steam pop or automatic shut off, discontinue RF energy. Remove the catheter for visual inspection and check for coagulum, charring, or other catheter defects.
Manual prebending of the distal curve may damage the steering mechanism and may cause patient injury. Do not attempt ablation with the catheter without the use of the DiamondTemp irrigation pump and DiamondTemp generator and approved accessories. Do not attempt ablation without the use of the irrigation pump. Before attempting ablation, make sure the pump flow rate is at the minimum continuous flow and the pump is actively communicating with the generator. The potential adverse events that may be associated with ablation procedures can vary greatly in frequency and severity and may necessitate additional medical intervention, including surgery. Carefully review the specific indications, contraindications, warnings, precautions, and adverse events included with each DiamondTemp catheter, prior to use of the DiamondTemp generator or irrigation pump. The irrigation pump is designed for use only with sterile heparinized normal saline solution. Specified flow-rate accuracy may not be maintained when used with incompatible fluids or delivery devices. The air bubble detector is disabled during irrigation pump priming and purging functions. Do not prime or purge the catheter when it is inserted in the vasculature of the patient. Do not remove the irrigation tubing set from the irrigation pump while the tubing set is in-line with a catheter that is inside the patient. The DiamondTemp Generator Connection Box E is to be used with the DiamondTemp Ablation system and Ensite™ cardiac mapping and navigation systems configured to use NavX navigation technology. Use with other systems has not been assessed and may compromise patient or operator safety. Do not connect the Generator Connection Box E to the Ensite™ Amplifier with both the primary and secondary connectors. Only connect using one of the connector options.
Potential Adverse Events or Potential Complications
Potential complications/adverse events from cardiac catheterization and ablation include, but are not limited to, the following: Abnormal vision; Air embolism; Anaphylaxis; Anemia; Aneurysm; Angina; Arrhythmia (including new or worsening of existing condition, or requiring cardioversion); Arterial or venous thrombus; Atrial septal defect; AV fistula; Cardiac arrest; Cardiac tamponade; Catheter entrapment leading to valve or heart wall damage; Catheter insertion site hematoma; Chest pain (nonspecific); Congestive heart failure exacerbation; Component damage to ICD or pacemaker; Coronary artery dissection; Death; Dislodgement of implantable device or permanent pacing lead; Dizziness; Embolic events, including infarction of other tissues, coronary, pulmonary, and bowel structures; Endocarditis; Esophageal damage or necrosis; Exacerbation of COPD; Exacerbation of pre-existing atrial fibrillation; Fluid overload; Gastroparesis or GI event; Hemorrhage; Hemothorax; Hypotension; Hypoxia; Inadvertent AV block; Infection; Myocardial infarction; Neck pain, back pain, or groin pain; Palpitations; Perforation (cardiac); Pericardial effusion; Pericarditis; Peripheral venous thrombosis; Phrenic nerve damage; Pleural effusion; Pneumonia; Pneumothorax; Pseudoaneurysm; Pulmonary edema; Pulmonary vein stenosis; Radiation injury resulting in dermatitis, erythema, etc.; Renal insufficiency or failure; Respiratory failure; Seizure; Sepsis; Skin burns; Stroke or cerebrovascular incident; Syncope; Thromboembolic event; Transient ischemic attack; Vasovagal reaction; Ventricular arrhythmia; Vessel wall or valvular damage or insufficiency.
See the appropriate product device manuals for detailed information regarding the RF ablation procedure, indications (or intended use), contraindications, warnings, precautions, and potential complications/adverse events. See the appropriate eIFUs for the DiamondTemp catheters, DiamondTemp RF generator, DiamondTemp irrigation pump, DiamondTemp irrigation tubing set, DiamondTemp catheter-to-RF generator cable, DiamondTemp GenConnect cable, EGM cable, and DiamondTemp Generator Connection Box E.
For further information, call Medtronic at 800-328-2518 and/or consult the Medtronic website at medtronic.com.
Caution: Federal law (USA) restricts these devices to sale by or on the order of a physician.
Back to top
FlexCath Contour steerable sheath
Intended Use
The FlexCath Contour steerable sheath facilitates introducing various cardiovascular catheters into the heart.
Indications for Use
The FlexCath Contour steerable sheath is indicated for percutaneous catheter, or transseptal needle introduction into the vasculature and into the chambers of the heart, including the left side of the heart through the interatrial septum. The sheath deflection facilitates device positioning.
Contraindications
The FlexCath Contour steerable sheath is contraindicated for placement in the left atrium or ventricle if:
- The patient has an intra-atrial septal patch or has had other surgical intervention in or adjacent to the intra-atrial septum.
- The patient has had a previous embolic event from the left side of the heart within two months of the procedure.
- The patient has known or suspected atrial myxoma.
Compatible Catheter Sizes:
FlexCath Contour Steerable Device (10 Fr)
The sheath can be used with Medtronic diagnostic and ablation catheter sizes from 7 Fr (2.3 mm) up to 9.5 Fr (3.2 mm).
FlexCath Contour Steerable Device (12 Fr)
The sheath can be used with Medtronic diagnostic and ablation catheter sizes from 7 Fr (2.3 mm) up to 10.5 Fr (3.5 mm).
Warnings/Precautions
This is a single use sheath to be used in a single patient. Do not resterilize this sheath for purpose of reuse. The dilator is compatible with transseptal needles that are at least 89 cm in length and less than 21 Gauge outer diameter. Do not use the sheath if it is kinked or damaged. Only physicians trained in left-sided catheterization should use this sheath during transseptal puncture. Administer appropriate levels of peri-procedural anticoagulation therapy for patients undergoing left-sided, right-sided, and transseptal cardiac procedures. Administer anticoagulation therapy during and post-procedure according to patient conditions and institutional standards. Introducing any catheter or sheath into the circulatory system entails the risk of air embolism, which can occlude vessels and lead to tissue infarction with serious consequences. To minimize the risk of air embolism, observe and remove any air prior to introducing the sheath and during the procedure. Do not advance the dilator or sheath through the interatrial septum without first confirming left atrial access to prevent advancing these components into an undesired location. Remove the guide wire and dilator from the sheath or insert the catheter into the sheath before slowly aspirating and flushing the sheath. Minimize catheter exchanges and always advance and withdraw catheters slowly. Follow advancement or withdrawal of catheters with appropriate aspiration and flushing according to institutional standards or consensus statements. Connect to a continuous drip to minimize back-bleeding. Do not pass the sheath through a prosthetic heart valve (mechanical or tissue). The sheath may become trapped in the valve, damaging the valve and causing valvular insufficiency or premature failure of the prosthetic valve. Cardiac catheterization procedures should be performed only in a fully equipped facility. This sheath should be used only by, or under the supervision of, physicians trained in cardiac catheterization procedures. Use extreme care when manipulating the sheath. Do not use excessive force to advance or withdraw the sheath, especially if resistance is encountered. Only physicians trained in left-sided catheterization should use this sheath during transseptal puncture.
Potential Adverse Events or Potential Complications
Potential adverse events associated with cannulation of the peripheral vasculature and intracardiac placement of the sheath and dilator may include the following conditions:
Access site complications (hematoma, infection, thrombosis, ecchymosis, AV fistula, bleeding from puncture site, hemorrhage); Air embolism; Arrhythmia (such as atrial fibrillation, atrial flutter, heart block requiring permanent pacemaker, ventricular tachycardia); Cardiac arrest; Chest discomfort, pain, or pressure; Coronary artery spasm; Damage to heart tissue or vasculature; Death; Endocarditis; Entrapment of the sheath within the patient; Hemothorax; Iatrogenic atrial septal defect (iASD); Infection (such as pericarditis, sepsis, urinary); Myocardial infarction; Perforation of venous, cardiac or surrounding tissue; Pericardial effusion, tamponade; Pericarditis; Pleural effusion; Pneumothorax; Pseudoaneurysm; Pulmonary edema; Pulmonary embolism; PV stenosis; Stroke; Thrombus; Transient ischemic attack (TIA); Valve damage; Vasovagal reaction
Refer to the device technical manual for detailed information regarding the procedure, indications, contraindications, warnings, precautions, and potential complications/adverse events. For further information, please call Medtronic at 800-238-2518 and/or consult the Medtronic website at medtronic.com.
Caution: Federal law (USA) restricts this device to sale by or on the order of a physician.
Back to top
FlexCath Cross transseptal solution
Prior to using these devices, please review the Instructions for Use for a complete listing of indications, contraindications, warnings, precautions, potential adverse events, and directions for use. FlexCath Cross™ devices are FDA cleared. Not all FlexCath Cross™ devices are CE marked.
Back to top
Previously known as AcQCross™ transseptal system. Anticipated rebranding to be complete summer 2023.
Freezor cardiac cryoablation catheter
Indications
The Freezor cardiac cryoablation catheter, CryoConsole system, and related accessories are indicated for the cryoablation of the conducting tissues of the heart in the treatment of adult and pediatric patients over two years of age, with atrioventricular nodal reentrant tachycardia (AVNRT).
Contraindications
This device is contraindicated in patients with active systemic infection, cryoglobulinemia, intracardiac mural thrombus, myxoma, interatrial baffle or patch.
Warnings/Precautions
Closely monitor AV conduction during cryo energy delivery near the AV node due to possible risk of complete atrioventricular (AV) block. Immediately terminate energy delivery if partial or complete AV block is noted. Cryoablation involving coronary vessels has been associated with subsequent clinically significant arterial stenosis. Care should be taken to minimize unnecessary contact with coronary vessels during cryoablation. Endotoxins may cause pyrogenic responses in patients including inflammation and fever which can result in anaphylactic shock and death. Caution should be used with regard to endotoxins in pediatric populations as standard endotoxin limits do not differentiate between adult and pediatric populations. Ethylene Oxide residuals remaining after sterilization are known to cause a potential increase in a number of biological effects including irritation, organ damage, mutagenicity and carcinogenicity. Calculations for ethylene oxide and ethylene chlorohydrin residuals indicate that the risk of these effects may be increased in patients under 3.8kg. Avoid catheter entanglement with other catheters devices or wires. Closely monitor patients undergoing cardiac ablation procedures during the post-ablation period for clinical adverse events. Do not re-use, reprocess or resterilize this device for purpose of reuse. This device is intended only to be used once for a single patient. The use of CryoMapping mode does not prevent injuries to the patient. CryoMapping mode is used to determine if the site is appropriate for ablation. The Freezor Catheter contains a pressurized refrigerant during operation. Release of this gas into the circulatory system due to equipment failure or misuse could result in gas embolism. Do not pull on the catheter, sheath, umbilical cables, or console while the catheter tip is frozen to the tissue, as this may lead to tissue injury. Always advance and withdraw components slowly to minimize the vacuum created and therefore minimize the risk of air embolism. Do not connect the Freezor Catheter to any radiofrequency generator or use the Freezor Catheter to deliver RF ablation energy. Before powering up an RF generator of applying RF energy, disconnect the cryoablation catheter from the CryoConsole. The use of fluoroscopy during catheter ablation procedures presents the potential for significant x-ray exposure to both patients and laboratory staff. Give careful consideration before using the device in pregnant women. Avoid positioning the catheter around the chordae tendineae, as this increases the likelihood of catheter entrapment within the heart. Do not pass the catheter through a prosthetic heart valve (mechanical or tissue).
Catheter handling:
- Use extreme care when manipulating the catheter. Lack of careful attention can result in injury such as perforation or tamponade.
- Do not use excessive force to advance, withdraw, the catheter, especially if resistance is encountered.
- Do not use the catheter if it is kinked, damaged, or cannot be straightened.
- Straighten the cooling segment before inserting or withdrawing the catheter.
- Do not at any time preshape or bend the catheter shaft or cooling segment.
- Catheter advancement should be performed under fluoroscopic guidance or other appropriate visualization technique.
Use only isolated equipment (IEC 60601-1 Type CF equipment, or equivalent) with the CryoConsole and catheters. The Medtronic cryoablation catheters, refrigerant tanks, and other Medtronic CryoConsole components should only be used with the Medtronic CryoConsole. If the sterile packaging or catheter is damaged, do not use the catheter. This equipment should be used only by physicians that perform cardiac ablation. Do not expose the catheter handle or coaxial and electrical connectors to fluids or solvents. Disconnect the catheter’s electrical connection prior to cardioversion/defibrillation. Administer appropriate levels of peri-procedural anticoagulation therapy for patients undergoing right-sided procedures. The Freezor catheter has not been evaluated for safety and compatibility in the magnetic resonance (MR) environment. Scanning a patient during the use of this device may result in patient injury.
Potential Complications
Potential complications that may be associated with cardiac catheterization and ablation listed alphabetically below include, but are not limited to: Access site complications including, hematoma, infection, thrombosis, ecchymosis, AV fistula, bleeding from puncture site, hemorrhage; arrhythmias (such as atrial fibrillation, atrial flutter, tachycardia); cardiac arrest and/or death; chest discomfort, pain or pressure; coronary artery spasm/stenosis; damage to heart tissue or vasculature; endocarditis; entrapment; heart block, requiring permanent pacemaker; hemothorax; infection; perforation of venous, cardiac or surrounding tissue, pericardial effusion, tamponade; pericarditis; phrenic nerve injury; pleural effusion; pneumothorax; pseudoaneurysm; pulmonary edema; pulmonary embolism; stroke; tissue infarction (such as myocardial infarction or renal infarction); thrombus; transient ischemic attack; vagal nerve injury (such as gastroparesis); vasovagal reaction.
Refer to the device technical manual for detailed information regarding the procedure, indications, contraindications, warnings, precautions, and potential complications/adverse events. For further information, please call Medtronic at 800-238-2518 and/or consult the Medtronic website at medtronic.com.
Caution: Federal law (USA) restricts these devices to sale by or on the order of a physician.
Back to top
Freezor MAX cardiac cryoablation catheter
Indications
The Freezor™ MAX cardiac cryoablation catheter is used as an adjunctive device in the endocardial treatment of paroxysmal and persistent atrial fibrillation (episode duration less than six months) in conjunction with the Arctic Front™ family of cardiac cryoablation catheters for the following uses: 1) Gap cryoablation to complete electrical isolation of the pulmonary veins 2) Cryoablation of focal trigger sites and 3) Creation of ablation line between the inferior vena cava and the tricuspid valve.
Contraindications
Use of Freezor™ MAX Cardiac Cryoablation Catheter is contraindicated in patients with active systemic infections, in patients with cryoglobulinemia, intracardiac mural thrombus, myxoma or interatrial baffle or patch. The Freezor MAX Cardiac Cryoablation Catheter is also contraindicated in patients with a body mass under 4.4 kg.
Warnings/Precautions
Do not reuse or reprocess this device for purpose of reuse. This catheter is intended only to be used once for a single patient. Do not connect the cryoablation catheter to a radiofrequency (RF) generator or use it to deliver RF energy. Disconnect the catheter’s electrical connection prior to cardioversion/defibrillation.
Catheter handling:
- Use extreme care when manipulating the catheter. Lack of careful attention can result in injury such as perforation or tamponade.
- Do not use excessive force to advance, withdraw the catheter, especially if resistance is encountered.
- Do not use the catheter if it is kinked, damaged, or cannot be straightened. Straighten the cooling segment before inserting or withdrawing the catheter.
- Do not at any time preshape or bend the catheter shaft or cooling segment.
- Catheter advancement should be performed under fluoroscopic guidance or other appropriate visualization technique.
Avoid positioning the catheter around the chordae tendineae. The catheter contains pressurized refrigerant during operation. Release of this gas into the circulatory system due to equipment failure or misuse could result in gas embolism. Do not pass the catheter through a prosthetic heart valve (mechanical or tissue). Use adequate fluoroscopic visualization or other appropriate visualization technique during a transaortic approach to avoid placing the ablation catheter within the coronary vasculature. Do not pull on the catheter, sheath, umbilical cables, or console while the catheter is frozen to the tissue, as this may lead to tissue injury. Always advance and withdraw components slowly to minimize the vacuum created. Closely monitor AV conduction during cryo energy delivery near the AV node due to possible risk of complete atrioventricular block.
Perform cryoablation procedures only within the environmental parameters. Do not expose the catheter handle or coaxial and electrical connectors to fluids or solvents. The use of fluoroscopy during catheter ablation procedures presents the potential for significant x-ray exposure to both patients and laboratory staff. Give careful consideration before using the catheter in pregnant women. Use only isolated equipment (IEC 60601-1 Type CF equipment, or equivalent) with the CryoConsole and catheters. The safety of the catheter in the MR environment is unknown (such as any heating, migration, or image artifacts). Scanning a patient during the use of this device may result in patient injury. Avoid catheter entanglement with other catheters, devices, or wires. Closely monitor patients undergoing cardiac ablation procedures during the post-ablation period for clinical adverse events. Before powering up an RF generator or applying RF energy, disconnect the cryoablation catheter from the CryoConsole. If the device is damaged or the integrity of the sterilization barrier has been compromised, do not use the product. The Medtronic cryoablation catheters, refrigerant tanks, and other Medtronic CryoConsole components should only be used with the Medtronic CryoConsole.
The Freezor MAX catheter was not studied for safety of changes in anticoagulation therapy in patients with paroxysmal atrial fibrillation. This equipment should be used only by physicians that perform cryoablation procedures. Cryoablation procedures should be performed only in a fully equipped facility.
Potential Complications
Potential complications/adverse events from cardiac catheterization and ablation include, but are not limited to the following: Access site complications including, hematoma, infection, thrombosis, ecchymosis, AV fistula, bleeding from the puncture site, hemorrhage; arrhythmias (such as atrial fibrillation, atrial flutter, tachycardia); cardiac arrest; chest discomfort, pain or pressure; coronary artery spasm/stenosis; damage to heart tissue or vasculature; death; endocarditis; entrapment; esophageal damage (such as atrio-esophageal fistula); heart block, potentially requiring implantation of a permanent pacemaker; hemothorax; infection; perforation of venous, cardiac or surrounding tissue, pericardial effusion, tamponade; pericarditis; phrenic nerve injury; pleural effusion; pneumothorax; pseudoaneurysm; pulmonary edema; pulmonary embolism; stroke; tissue infarction (such as myocardial infarction or renal infarction); thrombus; transient ischemic attack; vagal nerve injury (such as gastroparesis); vasovagal reaction.
Refer to the device technical manual for detailed information regarding the procedure, indications, contraindications, warnings, precautions, and potential complications/adverse events. For further information, please call Medtronic at 800-238-2518 and/or consult the Medtronic website at medtronic.com.
Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician.
Back to top
Freezor Xtra cardiac cryoablation catheter
Indications
The Freezor Xtra cardiac cryoablation catheter, CryoConsole system, and related accessories are indicated for the cryoablation of the conducting tissues of the heart in the treatment of adult and pediatric patients over two years of age with atrioventricular nodal reentrant tachycardia (AVNRT). The Freezor Xtra catheter is also intended for minimally invasive cardiac surgery procedures, including surgical treatment of cardiac arrhythmias. The Freezor Xtra catheter freezes the target tissue and blocks the electrical conduction by creating an inflammatory response or cryonecrosis.
Contraindications
The Freezor Xtra Cardiac Cryoablation Catheter is contraindicated in patients with the following conditions:
- Active systemic infections
- Cryoglobulinemia
- Intracardiac mural thrombus
- Myxoma
- Interatrial baffle or patch
Warnings/Precautions
Do not reuse, reprocess, or resterilize this catheter for purpose of reuse. This catheter is intended only to be used once for a single patient. Closely monitor AV conduction during cryo energy delivery near the AV node due to possible risk of complete atrioventricular (AV) block. Immediately terminate energy delivery if partial or complete AV block is noted. The use of CryoMapping mode does not prevent injuries to the patient. CryoMapping mode is used to determine if the site is appropriate for ablation. The catheter contains pressurized refrigerant during operation. Release of this gas into the body or circulatory system due to equipment failure or misuse could result in gas embolism, pericardial tamponade, tissue emphysema, or other patient injury. Always advance and withdraw components slowly to minimize the vacuum created and therefore minimize the risk of air embolism. Endotoxins may cause pyrogenic responses in patients including inflammation and fever which can result in anaphylactic shock and death. Caution should be used with regard to endotoxins in pediatric populations as standard endotoxin limits do not differentiate between adult and pediatric populations. Ethylene Oxide residuals remaining after sterilization are known to cause a potential increase in a number of biological effects including irritation, organ damage, mutagenicity, and carcinogenicity. Calculations for ethylene oxide and ethylene chlorohydrin residuals indicate that the risk of these effects may be increased in patients under 3.8kg. Avoid catheter entanglement with other catheters, devices, or wires. Closely monitor patients undergoing cardiac ablation procedures during the post-ablation period for clinical adverse events. Do not pull on the Freezor Xtra catheter, sheath, umbilical cables, or console while the catheter is frozen to the tissue, as this may lead to tissue injury. Do not connect the Freezor Xtra catheter to a radiofrequency (RF) generator or use it to deliver RF ablation energy. Before powering up an RF generator or applying RF energy, disconnect the cryoablation catheter from the CryoConsole. The use of fluoroscopy during ablation procedures presents the potential for significant x-ray exposure to both patients and laboratory staff. Give careful consideration before using the device in pregnant women.
Catheter handling:
- Use extreme care when manipulating the catheter. Lack of careful attention can result in injury such as perforation or tamponade.
- Do not use excessive force to advance or withdraw the catheter, especially if resistance is encountered.
- Do not use the catheter if it is kinked, damaged, or cannot be straightened.
- Straighten the cooling segment before inserting or withdrawing the catheter.
- Do not at any time preshape or bend the catheter shaft or cooling segment.
- Catheter advancement should be performed under fluoroscopic guidance or other appropriate visualization technique.
Use only isolated equipment (IEC 60601-1 Type CF equipment, or equivalent) with the CryoConsole and catheters. The Freezor Xtra catheter has not been evaluated for safety and compatibility in the magnetic resonance (MR) environment. Scanning a patient during the use of this device may result in patient injury. The Medtronic cryoablation catheters, refrigerant tanks, and other Medtronic CryoConsole components should only be used with the Medtronic CryoConsole. If the sterile packaging or catheter is damaged, do not use the catheter. Do not expose the catheter handle or coaxial and electrical connectors to fluids or solvents. Disconnect the catheter’s electrical connection prior to cardioversion/defibrillation. Cryoablation involving coronary vessels with liquid nitrous oxide systems has been associated with subsequent clinically significant arterial stenosis. Care should be taken to minimize unnecessary contact with coronary vessels during cryoablation.
Avoid positioning the catheter around the chordae tendineae, as this increases the likelihood of catheter entrapment within the heart. Do not pass the catheter through a prosthetic heart valve (mechanical or tissue). Administer appropriate levels of peri-procedural anticoagulation therapy for patients undergoing endocardial right-sided procedures. This equipment should be used only by physicians that perform cardiac ablation.
Potential Complications
Potential complications that may be associated with cardiac catheterization and ablation listed alphabetically below include, but are not limited to: Access site complications including, hematoma, infection, thrombosis, ecchymosis, AV fistula, bleeding from puncture site, hemorrhage; arrhythmias (such as atrial fibrillation, atrial flutter, tachycardia); cardiac arrest and/or death; chest discomfort, pain or pressure; coronary artery spasm/stenosis; damage to heart tissue or vasculature; endocarditis; entrapment; heart block, requiring permanent pacemaker; hemothorax; infection; perforation of venous, cardiac or surrounding tissue, pericardial effusion, tamponade; pericarditis; phrenic nerve injury; pleural effusion; pneumothorax; pseudoaneurysm; pulmonary edema; pulmonary embolism; stroke; tissue infarction (such as myocardial infarction or renal infarction); thrombus; transient ischemic attack; vagal nerve injury (such as gastroparesis); vasovagal reaction.
Refer to the device technical manual for detailed information regarding the procedure, indications, contraindications, warnings, precautions, and potential complications/adverse events. For further information, please call Medtronic at ;800-238-2518 and/or consult the Medtronic website at medtronic.com.
Caution: Federal law (USA) restricts this device to sale by or on the order of a physician.
Back to top
Nitron CryoConsole cardiac cryoablation system
Indications (or Intended Use)
The Nitron CryoConsole (the console), together with its components and specified catheters, is for use in performing cardiac ablation procedures.
Contraindications
See Technical Manual for the compatible Medtronic catheter being used.
Warnings and Precautions
These warnings apply in general to using the Nitron CryoConsole for cardiac cryoablation. Refer to the technical manual for console-compatible catheters for more information related to catheter use. • Connected EP lab equipment – Any equipment (e.g. EP recording system, HDMI, etc.) connected to the upper rear connection panel or to the auto connection box ECG cable receptacle of the device shall be IEC 60601-1 compliant or have a 60601-1-compliant isolation transformer on the interconnect cable between the Nitron CryoConsole and the EP lab equipment. • Electric shock – To avoid risk of electric shock, this equipment must only be connected to a supply main with protective earth. • Cryoadhesion – Do not pull on the catheter, sheath, umbilical cables, or console while the catheter is frozen to the tissue, as this may lead to tissue injury. • Improper connection – Do not connect a cryoablation catheter to a radiofrequency (RF) generator or use it to deliver RF energy. Doing this may cause catheter malfunction or patient harm. • Other accessories, transducers, and cables – Use of accessories, transducers and cables other than those specified or provided by the manufacturer of this equipment could result in increased electromagnetic emissions or decreased electromagnetic immunity of this equipment and result in improper operation.
The console meets the requirements of IEC 60601-1. It is the user’s responsibility after installation to verify and ensure that the console meets the applicable local electrical safety requirements. Perform cryoablation procedures only within the environmental parameters. Cryoablation procedures should be performed only in an electrophysiology facility by or under the supervision of physicians trained in cryoablation. Do not modify this equipment. Modifications may reduce system effectiveness and impact patient health. The system must be installed by a qualified/trained Medtronic representative. All information technology equipment that is attached to the console must be approved by a third party to the requirements of UL 1950 or EN 60950. Minimize exposure to nitrous oxide to prevent short-term behavioral and long-term reproductive health effects. The console and its accessories have not been evaluated for safety and compatibility in the magnetic resonance (MR) environment and are unknown which may lead to patient injury. Use only compatible Medtronic cryoablation catheters and components and Medtronic-provided refrigerant tanks with the console. The safety and use of other catheters or components has not been tested.
safety and compatibility in the magnetic resonance (MR) environment and are unknown which may lead to patient injury. Use only compatible Medtronic cryoablation catheters and components and Medtronic-provided refrigerant tanks with the console. The safety and use
Potential Adverse Events or Potential Complications
Potential adverse events associated with console procedures include, but are not limited to, the following conditions: • Electric shock • Foreign body/biological reaction • Long-term reproductive issues • Thermal injury
Refer to the device technical manual for detailed information regarding the procedure, indications, contraindications, warnings, precautions, and potential complications/adverse events. For further information, please call Medtronic at 800-328-2518 and/or consult the Medtronic website at medtronic.com.
Caution:Federal law (USA) restricts these devices to sale by or on the order of a physician.
Back to top
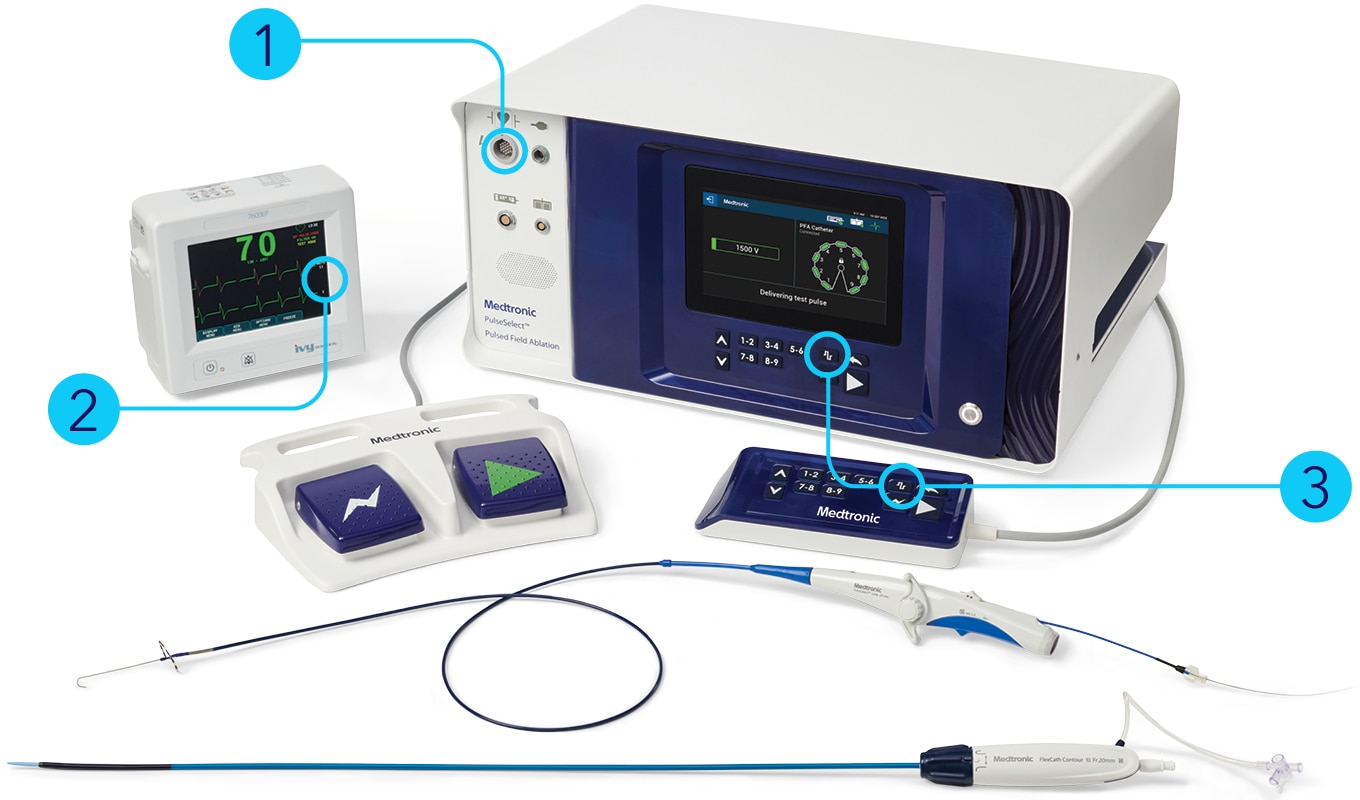
PulseSelect™ pulsed field ablation (PFA) system
Indications (or Intended Use)
The PulseSelect™ Pulsed Field Ablation (PFA) System is indicated for use in cardiac electrophysiological mapping (stimulation and recording) and for treatment of drug refractory, recurrent, symptomatic paroxysmal atrial fibrillation or persistent atrial fibrillation (episode duration less than 1 year).
Contraindications
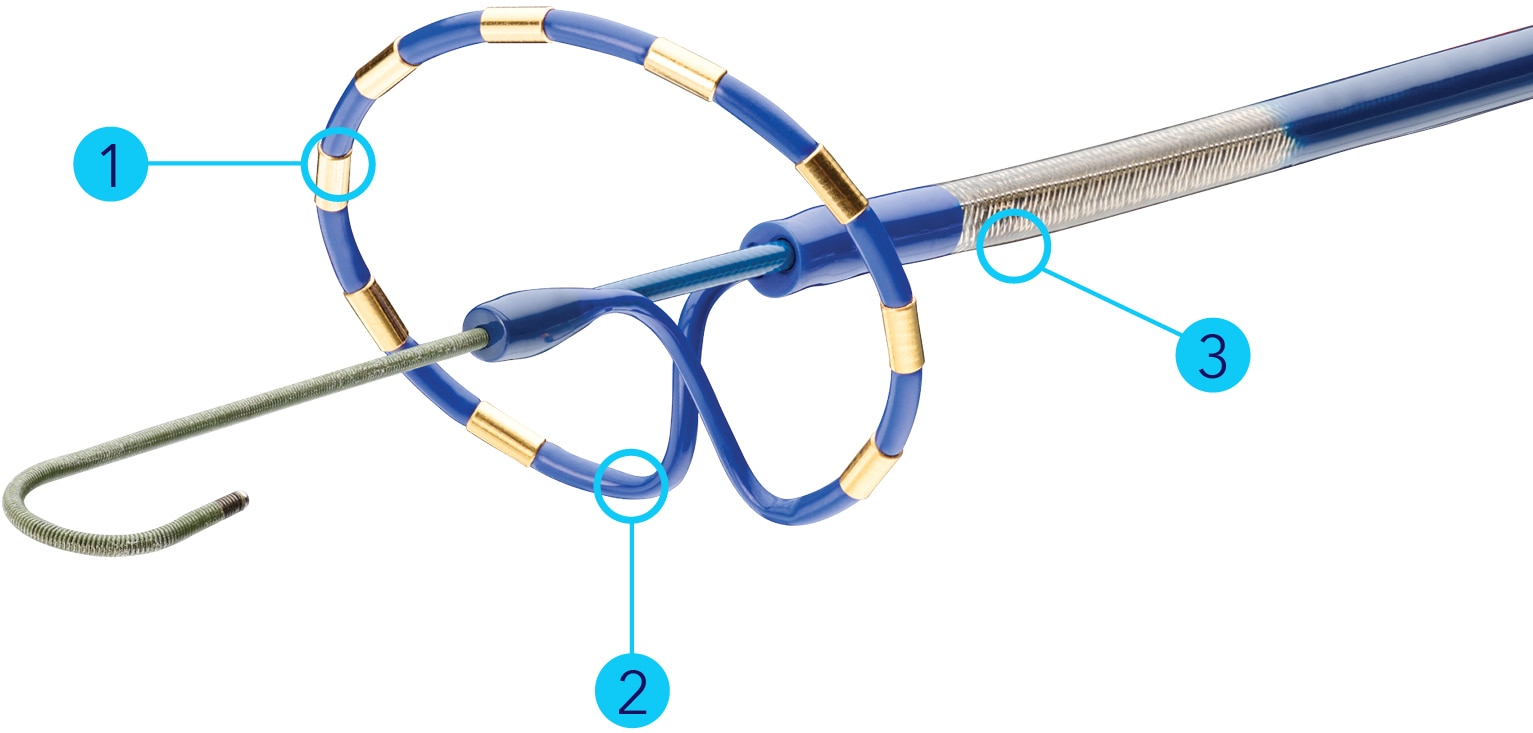
The PulseSelect PFA loop catheter is contraindicated for use in patients with the following conditions:
• Active systemic infections
• A known sensitivity to Heparin
• Blood clotting abnormalities
• Permanently implanted metallic objects in the left atrium
The catheter is also contraindicated in conditions where the manipulation of the catheter within the heart would be unsafe, such as intracardiac mural thrombus.
The catheter is not recommended for use in patients who cannot undergo standard anticoagulation protocol for a left-sided cardiac procedure, or who have had a recent coagulopathy or embolic event.
Warnings and Precautions
To reduce the possibility of hazards associated with use of the PulseSelect PFA loop catheter: • Use the catheter only in the recommended anatomical location. • Maintain the catheter position during the ablation. • If coughing occurs, reposition the catheter more proximally and review sedation management. • Ensure electrodes are not in contact with any metal during ablations (for example the guide wire). • Maintain substantially circular array to ensure uniform field distribution. • If the electrode array is deployed to deliver ablation energy, avoid continuing to move the slide control forward to prevent the guide wire lumen from coming too close to the electrode array.
It is recommended that the array be captured while it is submerged to help reduce the possibility of air becoming entrapped around the electrode array during capture and catheter insertion.
Catheter integrity – Use care to avoid damage to the catheter. • Do not bend or kink the leading end of the catheter. Doing so could cause damage to the catheter lumen and make it unusable. • Monitor the catheter throughout the procedure. If a flash is observed in the luer, replace the catheter immediately.
Electrode-electrode contact – Avoid contact between electrodes. Contact between electrodes may create a short circuit.
Embolism risk – Introducing any catheter or sheath into the circulatory system entails the risk of air, gas, or thromboembolism, which can occlude vessels and lead to tissue infarction with serious consequences. • Avoid unnecessary catheter exchanges to minimize sheath-related embolic events. • Always advance and withdraw components slowly to minimize the vacuum created and the risk of air embolism. • Aspirate and flush the sheath frequently to help minimize the potential for embolic events resulting from the introduction of air or clot formation within the sheath.
Fluoroscopy use during catheter placement – Only perform catheter ablation after giving adequate attention to the potential radiation exposure associated with the procedure, and taking steps to minimize this exposure. Give careful consideration before using the device in pregnant women.
For single use only – The PFA catheter is intended only to be used once for a single patient. Do not reuse, reprocess, or resterilize the PFA catheter.
Careful manipulation of the catheter is necessary to avoid cardiac damage, perforation, or tamponade. • Do not use excessive force to advance, withdraw, or rotate the catheter, especially if resistance is encountered. Excessive force may lead to catheter damage and blood loss. • Use imaging guidance during catheter advancement, manipulation, and placement. • Vascular perforation is an inherent risk of catheter placement. • Performing ablation with the PFA catheter array inside the sheath may result in damage to the array or the sheath and should be avoided. • Performing steering manipulation with the PFA catheter array inside the sheath may result in damage to the catheter steering mechanism or the sheath and should be avoided. • The PFA generator is capable of delivering significant energy. Do not touch the ablation electrodes of the PFA catheter while operating the generator. • If the system is to be tested outside of the body, the electrode array must be immersed in saline solution in a plastic container. Never test PFA delivery in direct contact with skin.
Use of imaging during catheter manipulation and placement is strongly advised. Manipulating the catheter without imaging may result in damage to cardiac and vascular structures.
Other devices, wires, or catheters – Avoid catheter entanglement with other devices, wires, or catheters, for example, intracardiac echo catheters. Failure to do so may increase the risk of entrapment of the array or damage to the array, which may affect retrieval of the device into the transseptal sheath.
Phrenic nerve injury – To reduce the potential for phrenic nerve injury, assess for proximity of the ablation catheter to the nerve using an appropriate technique such as pacing for local phrenic nerve capture or using the test pulse feature before ablation. Stop ablation immediately if phrenic nerve impairment is observed and assess for injury.
Sheath and guide wire required – Do not attempt to advance or withdraw the catheter through the vasculature without the use of a sheath and guide wire, as it may result in damage to cardiac and vascular structures.
Implanted devices, such as pacemakers and implantable cardioverter-defibrillators (ICDs), may be adversely affected by PFA energy. • Keep external sources of pacing and defibrillation available during ablation. • Program pacemaker sensing parameters to asynchronous pacing to ensure that PFA energy is not sensed as an intrinsic event. • Deactivate ICD detection during the delivery of PFA energy. • Perform complete implantable device testing before and after ablation. • Monitor surface and intracardiac electrograms or vital signs during PFA energy delivery to assess for device interaction. Take appropriate action if any interaction is detected. • Refer to the appropriate implantable device technical manual for additional information.
Electrical safety requirements – The PFA generator meets the requirements of IEC 60601-1. It is the user’s responsibility after installation to verify and ensure that the generator meets the applicable local electrical safety requirements.
Electric shock – To avoid risk of electric shock, this equipment must only be connected to a supply main with protective earth.
Electromagnetic interference (EMI) radiated – The generator emits energy during ablation at a frequency level that may cause EMI with unshielded electronic equipment. To minimize EMI, the generator should be moved away from any other electronic device. If EMI is apparent during the application of energy, EMI may be reduced by repositioning the generator or other equipment.
Electromagnetic interference (EMI) susceptibility – The generator has been designed to minimize electromagnetic interference (EMI). If interference should occur, move the generator away from the device generating the interference or place the generator at a different angle.
Leakage current from connected devices – Use only isolated equipment (IEC 60601-1 Type CF equipment, or equivalent) with the PFA system and catheters or patient injury or death may occur.
Potential Adverse Events or Potential Complications
Potential adverse events associated with cardiac catheter ablation procedures include, but are not limited to, the following conditions: • Access site complications (such as, bruising, ecchymosis, arteriovenous fistula, hematoma, pseudoaneurysm) • Anemia • Arrhythmias, proarrhythmia (such as, atrial flutter, bradycardia, heart block, tachycardia) • Bleeding, possibly requiring transfusion • Bruising • Cardiopulmonary arrest • Perforation of the heart or other organs during transseptal puncture or other procedures • Cardiac tamponade • Catheter entrapment in cardiac structures requiring intervention • Cerebrovascular accident [such as stroke, transient ischemic attack (TIA)] • Chest discomfort, pain, or pressure • Collateral damage to the conduction system or coronary vasculature • Cough • Death • Embolism • Esophageal damage (including atrial esophageal fistula) • Hemoptysis • Hypotension • Hypertension • Infections (such as, sepsis) • Myocardial infarction or ischemia • Nerve injury or nerve damage (for example phrenic nerve injury) • Pericarditis or endocarditis • Pericardial effusion • Pneumothorax • Pulmonary edema • Pulmonary vein dissection • Pulmonary vein stenosis • Radiation injury or damage and late malignancy • Skin laceration or puncture • Sore throat • Unintended complete or incomplete atrioventricular node (AV-Node) or sinus node block or damage • Valvular insufficiency or damage.
Refer to the device technical manual for detailed information regarding the procedure, indications, contraindications, warnings, precautions, and potential complications/adverse events. For further information, please call Medtronic at 1-800-328-2518 and/or consult the Medtronic website at www.medtronic.com.
Caution: Federal law (USA) restricts these devices to sale by or on the order of a physician.
Back to top
MAJ_79177