KYPHON ASSIST™ Directional Cannula for Balloon Kyphoplasty
The Kyphon Assist™ directional cannula provides more predictable inflation and direction of cavity creation during balloon kyphoplasty surgery for vertebral compression fractures. Available in both 8 ga and 10 ga.
DOWNLOAD THE HCP GUIDEOverview
GET TO KNOW KYPHON ASSIST™
Watch the video introduction to the Kyphon Assist™ directional cannula.
KEY BENEFITS
MORE CONTROL AND MORE HEIGHT
When compared to standard access cannulas,1 the Kyphon Assist™ can redirect 76% of the inflatable bone tamp volume and increase the directional height by 37% for more height restoration potential.
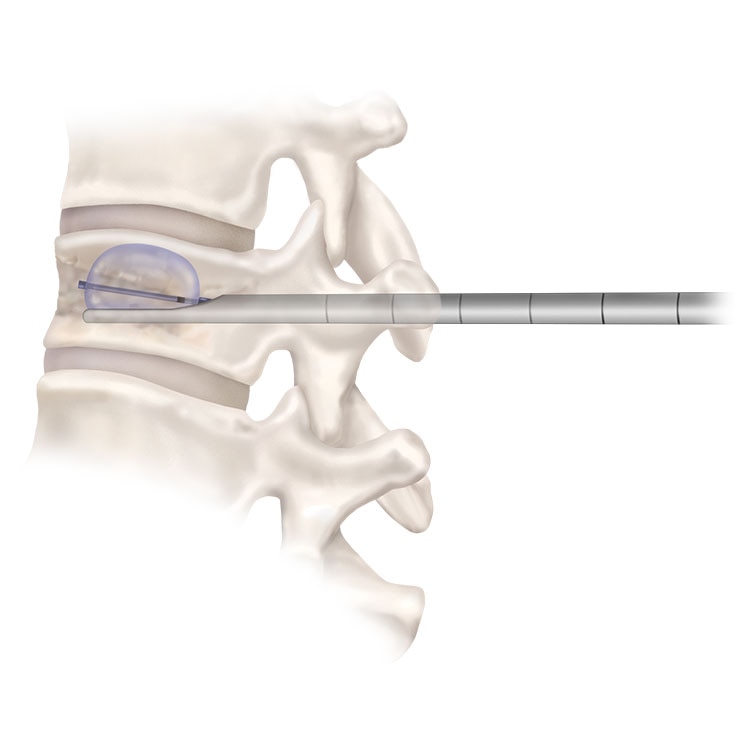
MORE FLEXIBILITY AND MORE CONFIDENCE
The Kyphon Assist™ can be used with any size of inflatable bone tamp allowing flexibility for procedural goals and anatomic considerations. It comes in the familiar One-Step™ access and can be exchanged as needed for more accurate balloon placement.
The Kyphon Assist™ also provides the ability to inflate away from the lateral walls and endplate defects providing more control and confidence in balloon kyphoplasty procedures.
Note: When using the Kyphon Assist™ 10 ga directional cannula (T34S) with the Kyphon Express 400 psi-rated IBTs, item numbers K15A (10 mm) and K15B (15 mm), the maximum inflation volumes are reduced by 1 cc for adjusted maximum inflation volumes of 3 cc (K15A) and 4 cc (K15B).
KEY BENEFITS
Study Results
A recent study2 of nondirectional balloon technique (n=51) and VCF patients treated with directional balloon technique (n=49) demonstrated the following outcomes for patients treated with a directional balloon technique:
- Significantly immediate and sustained kyphotic angle correction post-surgery
- Significantly early and immediate outcome improvement: The improvement in VAS, ODI, and RMD scores is significantly better than nondirectional technique at 3 days.

COMPARISON OF THE CLINICAL OUTCOMES RESULTS BETWEEN 2 GROUPS BEFORE AND AFTER OPERATION
|
|
|
Preoperative |
3 days |
1 month |
3 months |
6 months |
12 months |
|---|---|---|---|---|---|---|---|
| VAS
|
Group 1 |
8.1 ± 1.2 |
4.5 ± 1.3*† |
2.4 ± 1.2* |
2.1 ± 1.1* |
1.9 ± 1.0* |
2.3 ± 1.2* |
| Group 2 |
8.0 ± 1.1 |
4.0 ± 1.1* |
2.2 ± 1.0* |
2.0 ± 1.0* |
1.8 ± 0.9* |
2.4 ± 1.3* |
|
| ODI
|
Group 1 |
48.1 ± 2.2 |
42.8 ± 3.2*† |
32.5 ± 2.2* |
25.4 ± 2.3* |
20.3 ± 2.1* |
18.8 ± 1.6* |
| Group 2 |
48.4 ± 1.9 |
34.2 ± 2.4* |
30.4 ± 1.99* |
24.3 ± 2.1* |
20.1 ± 1.9* |
18.6 ± 1.3* |
|
| RMD
|
Group 1 |
21.5 ± 1.8 |
12.3 ± 1.3*† |
7.2 ± 2.1* |
6.9 ± 2.5* |
5.7 ± 2.3* |
7.1 ± 2.0* |
| Group 2 |
21.0 ± 1.7 |
11.5 ± 1.2* |
6.7 ± 2.2* |
6.3 ± 2.3* |
5.5 ± 2.1* |
6.6 ± 1.9* |
Data were presented as mean ± standard deviation. ODI = Oswestry Disability Index. RMD = Roland Morris Disability Questionnaire. VAS = Visual Analogue Scale.
- Significantly reduced cement leakage rate: The directional balloon technique reduced cement leakage by 65% compared to conventional balloon kyphoplasty.
- Significantly shortened operative and exposure time:
- The directional balloon technique reduced operation time by 8% when compared to conventional balloon kyphoplasty.
- The directional balloon technique reduced X-ray exposure by 20% when compared to conventional balloon kyphoplasty.
| Group 1 Conventional BKP |
Group 2 Directional BKP |
P | |
|---|---|---|---|
|
Case (N) |
51 |
49 |
- |
|
Operation time (min.) |
47.8 ± 6.1 |
43.8 ± 5.7 |
.001 |
|
Times of x-ray exposure |
10 ± 2.0 |
8 ± 1.9 |
< .01 |
|
Blood loss (mL) |
2.4 ± 1.1 |
2.3 ± 1.0 |
.63 |
|
Inject cement volume (mL) |
4.6 ± 1.4 |
4.8 ± 1.3 |
.46 |
|
Bone cement leakage rate (n/N) |
12/51 (23.5%) |
4/49 (8.2%) |
.036 |
|
Leakage type |
|||
|
B |
1/51 |
1/49 |
.74 |
|
C |
9/51 |
2/49 |
.03 |
|
S |
3/51 |
1/49 |
.64 |
Quantitative data were presented as mean ± deviation.
METHODS
- The study compared non-Medtronic instruments.
- In group 1, a conventional balloon kyphoplasty technology was used (Percutaneous Kyphoplasty System, Shandong Dragon Crown Medical Co., Ltd) and in group 2, a new type of directional balloon dilation technology (Directional Percutaneous Kyphoplasty System, Suzhou and Science & Technology Development Co., Ltd) was used.
- Data are presented as means ± standard deviation. KA=Kyphosis angle, WA=Wedge-shaped angle.
- Significant kyphotic angle correction at 3 days could be attributed for early and immediate outcome improvement.
- Data were presented as mean ± standard deviation:
- ODI =Oswestry Disability Index
- RMD = Roland Morris Disability Questionnaire
- VAS = Visual Analogue Scale
- Study featured a small sample size and short follow up time. Further research is recommended to determine strength comparison between the controllable directional device, bone and balloon.
- Bone cement leakage was classified into 3 types:
- Type B: Cement leakage along the vertebrobasilar vein to the posterior border of the vertebral body.
- Type C: Cement leakage mainly along the cortical defect to the intervertebral disc.
- Type S: Cement leakage mainly around the vertebral body along the intervertebral vein.
RESOURCES
MARKETING TOOLS FOR YOUR PRACTICE
Access ready-made marketing and patient education tools.
GET TOOLSComparison between postoperative and preoperative in each group, P<05.
Group 1 compared with group 2 at different time points, P<.05.
Medtronic data on file. ETR31101239.
Wang P, Li J, Song Z, Peng Z, Wang G. Utilization of the directional balloon technique to improve the effectiveness of percutaneous kyphoplasty in the treatment of osteoporotic vertebral compression fractures and reduction of bone cement leakage. Medicine (Baltimore). 2019;98(19):e15272.
